Here’s what you need to know:
South African health care workers were given the Johnson & Johnson vaccine at a hospital in Johannesburg in March.Credit…Joao Silva/The New York Times
South Africa will resume the use of the Johnson & Johnson vaccine to inoculate health care workers next week, offering some relief to the country that has suffered a series of blows to its vaccination efforts in recent months, according to South African authorities.
The country suspended an early-access Johnson & Johnson vaccination program last week after health officials in the United States put a pause on the vaccine amid concerns of rare blood clots that emerged in a handful of people who received it.
South Africa’s decision to move forward again was the second green light this week for Johnson & Johnson. On Tuesday, the European Union drug regulator also recommended resuming the rollout of the company’s vaccine.
Now, many eyes are on Washington, where a federal advisory panel is scheduled to meet Friday to discuss whether to lift the pause in the United States.
The blood clots that led to the Johnson & Johnson suspensions were all reported in the United States. In South Africa, officials confirmed Thursday that no cases of clots have been reported among the roughly 290,000 health care workers who have received the vaccine so far.
“The temporary suspension in South Africa was in line with government’s commitment to ensure comprehensive measures are undertaken regarding vaccine rollout,” Khumbudzo Ntshavheni, a cabinet minister, told reporters on Thursday.
Health experts welcomed the resumption of the vaccine campaign in South Africa, which has recorded more coronavirus cases than any other country on the continent and has suffered serious setbacks in its attempt to combat the virus in recent months.
In February, health officials scrapped plans to use the AstraZeneca vaccine after it proved ineffective against a variant of the virus now dominant in South Africa. The decision came a week after a million doses of the vaccine arrived in the country and amid a devastating second wave of virus cases.
Though the Johnson & Johnson vaccine has not yet been approved for general use in South Africa, it has been used as part of a research study offering early access to the vaccine to the country’s 1.2 million health care workers.
South African health officials are gearing up to extend vaccinations to the general public starting in May. In a first step to launching a national rollout, the country last week opened its vaccine registration to people over 60 years old, who will be among the first to be inoculated.
That plan depends on tens of millions of doses of the Pfizer-BioNTech vaccine, which requires two doses and will be used in major cities. The single-shot Johnson & Johnson vaccine, which is easier to store and better for hard-to-reach populations, will be used in the country’s rural areas.
| United States › |
United StatesOn Apr. 21 |
14-day change |
| New cases |
64,853 |
–4% |
|
| New deaths |
879 |
–1% |
|
| World › |
WorldOn Apr. 21 |
14-day change |
| New cases |
952,928 |
+23% |
|
| New deaths |
17,951 |
+14% |
|
U.S. vaccinations ›

 People waiting in line to register for a vaccination in Brooklyn earlier this month.Credit…Spencer Platt/Getty Images
People waiting in line to register for a vaccination in Brooklyn earlier this month.Credit…Spencer Platt/Getty Images
Federal health officials appear to be leaning toward lifting their recommended pause on the use of Johnson & Johnson’s coronavirus vaccine after finding only a limited number of additional cases of a rare blood clotting disorder among recipients.
Instead, the Food and Drug Administration is likely to attach a warning to the vaccine’s label to inform health practitioners — and the public — about the exceedingly uncommon, but dangerous possible side effect.
Federal health officials are waiting to act until they hear from a committee of outside experts who advise the C.D.C. The committee is scheduled to meet on Friday to discuss whether to recommend lifting, extending or modifying the pause that was initiated on April 13.
“We know that it’s not a good thing to leave the pause going for any longer than it absolutely has to go for,” Dr. Peter Marks, the Food and Drug Administration’s top vaccine regulator, said Thursday, adding that a protracted pause could contribute to greater vaccine hesitancy. “Once, essentially, the adequate discussion has occurred, we’re prepared to move as quickly as we possibly can.”
When top federal health officials abruptly decided early last week to recommend a temporary halt in the use of the shot, six women had been reported to have suffered from the disorder, a combination of clots in the brain that led to bleeding and low platelets, components of the blood that normally help to heal wounds.
That was fewer than one in a million recipients of Johnson & Johnson’s shot in the United States. But officials worried that more cases were hidden or could develop shortly as the new vaccine rolled out.
That fear has not materialized.
Dr. Marks and Dr. Janet Woodcock, the F.D.A.’s acting commissioner, said the clotting disorder appeared to be nearly as rare as they hoped it would be when they recommended the pause.
“We’ve now received more cases, but it isn’t an avalanche,” Dr. Woodcock said “We’re not seeing a big surge, which is a great relief.”
Even if the C.D.C.’s advisory committee decides Friday that the benefits of Johnson & Johnson’s single-dose vaccine outweigh its risks, the company will still face manufacturing hurdles at a Baltimore plant that regulators have refused so far to certify. That plant was supposed to deliver the bulk of the nearly 100 million doses the firm had promised to have ready by the end of May.
But it would mean a temporary surge of about 10 million shots that were effectively put on hold when the pause was announced.
 A man who died of complications from the coronavirus was being cremated in Mumbai on Wednesday.Credit…Atul Loke for The New York Times
A man who died of complications from the coronavirus was being cremated in Mumbai on Wednesday.Credit…Atul Loke for The New York Times
India’s rapidly worsening coronavirus outbreak is now expanding on a scale beyond any previously measured in more than a year of the pandemic: The health ministry reported more than 310,000 new infections on Thursday, the most recorded in any country on a single day.
India’s total eclipsed the previous one-day high of 300,669 recorded coronavirus cases, set in the United States on Jan. 8, according to a New York Times database, though differences in testing levels from country to country, and a widespread lack of tests early in the pandemic, make comparisons difficult.
Over the past two months, the outbreak in India has exploded, with reports of superspreader gatherings, oxygen shortages and ambulances lined up outside hospitals because there were no ventilators for new patients.
As cases worldwide reach weekly records, a substantial proportion of the new infections are coming in India, a sobering reminder that the pandemic is far from over, even as infections decline and vaccinations speed ahead in the United States and other wealthy parts of the world. India has surpassed 15.6 million total reported infections so far, second-most after the United States.
The death toll has also begun to climb precipitously.
On Thursday, the Indian government recorded 2,104 deaths, and an average of more than 1,600 people have died of the virus every day for the past week. That is less than the tolls at the worst points of the pandemic in the United States or Brazil, but it is a steep increase from just two months ago, when fewer than 100 people in India were dying daily.
There are signs that the country’s health system, patchy even before the pandemic, is collapsing under the strain. On Tuesday, at least 22 people died in an accident in the central city of Nashik when a leak in a hospital’s main oxygen tank cut the flow of oxygen to Covid-19 patients.
The picture is staggeringly different from early February, when India was recording an average of just 11,000 cases a day, and domestic drug companies were pumping out millions of vaccine doses. More than 132 million Indians have received at least one dose, but supplies are running low and experts warn that the country is unlikely to meet its goal of inoculating 300 million people by the summer.
Critics say Prime Minister Narendra Modi, who imposed a harsh nationwide lockdown in March 2020 in the early stages of the pandemic, failed to prepare for a second wave or to warn Indians to remain vigilant against the virus, especially as more infectious variants began to spread.
Mr. Modi’s Hindu nationalist government has also allowed a massive Hindu festival to take place, drawing millions of pilgrims to the banks of the Ganges River, and his party has held packed political rallies in several states.
“India’s rapid slide into this unprecedented crisis is a direct result of complacency and lack of preparation by the government,” Ramanan Laxminarayan, the director of the Center for Disease Dynamics, Economics and Policy in Washington, wrote in The New York Times on Tuesday.
The hardest-hit region is Maharashtra, a populous western state that includes the financial hub of Mumbai. On Wednesday, the state’s top leader ordered government offices to operate at 15 percent capacity and imposed new restrictions on weddings and private transportation to slow the spread of the virus.
This week, Britain’s prime minister, Boris Johnson, and Japan’s prime minister, Yoshihide Suga, called off plans to visit India. On Thursday, the Australian prime minister, Scott Morrison, said that direct flights from India would be reduced by about 30 percent, and that Australians would be allowed to travel to India only in “very urgent circumstances.” Canada also suspended all direct flights from India and Pakistan starting Thursday night for 30 days.
 People relaxed in the Place des Vosges in central Paris on Saturday. Prime Minister Jean Castex said that France will relax many of its coronavirus restrictions in May. Credit…Dmitry Kostyukov for The New York Times
People relaxed in the Place des Vosges in central Paris on Saturday. Prime Minister Jean Castex said that France will relax many of its coronavirus restrictions in May. Credit…Dmitry Kostyukov for The New York Times
The French government outlined plans on Thursday to gradually reopen the country starting in early May, stoking hopes that life might finally return to something close to normal after more than a year of on-and-off pandemic restrictions.
Prime Minister Jean Castex said at a news conference that primary school students would be allowed to return to classrooms on Monday, followed by middle and high school students the following week. Travel restrictions will be lifted on May 3.
Depending on how things are going at that point, Mr. Castex said, retail stores, outdoor dining, and certain cultural and sporting activities could start to reopen in mid-May.
The pandemic situation appears to be improving in France, with the daily average number of new cases falling to about 32,000 from 42,000 the week before. Hospitalizations seem to have plateaued at nearly 6,000.
“The peak of the third wave seems to be behind us,” Mr. Castex said.
The government is hoping to alleviate the deep sense of pandemic fatigue that has taken root in France. When the country went into its third lockdown at the start of April, once again closing schools and “nonessential” retail stores, the move was met with anger and some pointed protests.
Hundreds of lingerie shops across France, closed under the lockdown order, have been mailing panties to Mr. Castex since the beginning of the week, as part of a campaign called “Action Culottée,” meaning “cheeky action,” which was coordinated on Facebook.
The country’s vaccination campaign, which stumbled for months, has gathered speed recently, and is now administering about 2.5 million doses a week. More than 13 million people have received at least one dose so far, and the country aims to raise the figure to 20 million — 30 percent of the population — by mid-May. Even so, France lags far behind countries like the United States, Britain and Israel in its vaccination efforts.
To limit the spread of highly transmissible virus variants, Mr. Castex said, France will tighten testing and quarantine requirements for travelers arriving from five countries — Brazil, Chile, Argentina, South Africa and India — where the variants are circulating widely.
 The Atlantic City boardwalk last July.Credit…Michelle Gustafson for The New York Times
The Atlantic City boardwalk last July.Credit…Michelle Gustafson for The New York Times
With summer on the horizon, states are beginning to rethink social-distancing measures.
In Rhode Island, Gov. Dan McKee said that starting May 7, the state will stop requiring masks outside, and social gatherings can increase to 25 people indoors and 75 people outdoors. By May 28, the state will lift capacity limits on businesses and houses of worship; the bar areas of restaurants will be able to open; and dance floors can once again be filled.
“It’s a good day for everyone here in the Ocean State,” Mr. McKee said at a news conference Thursday. “It’s a little early to put a ‘Mission Accomplished’ sign up but we’re getting ready to order that sign.”
Mr. McKee attributed the reopening plans to the state’s vaccination rate — 48 percent of residents have received at least one shot and 33 percent are fully vaccinated, according to a New York Times database. But masks will still be required indoors.
Rhode Island is not alone.
On Monday, Gov. Ned Lamont of Connecticut announced that his state would phase out all pandemic restrictions, except the indoor mask mandate, by May 19. And in New Jersey, Gov. Phil Murphy said Wednesday that he would announce “a pretty significant amount of guidance” for summer activities next week.
“We don’t want to lurch, in other words go forward and then have to pull something back,” Mr. Murphy said at his weekly news conference. “And we don’t want to start that now. But we also owe people our best guesses for what it’s going to look like for graduation, summer, the beaches and what not.”
As more people get vaccinated and the outdoors become more appealing with spring weather and sunshine, one question persists: Do we still need to wear masks outside? Science shows that the risk of viral transmission outside is very low. The Times’ Well columnist, Tara Parker-Pope, suggests making sure your activity meets two out of the following three conditions: outdoors, distanced and masked.
Global Roundup
 Police officers stood guard in Berlin as Germans demonstrated against coronavirus measures on Wednesday.Credit…Christian Mang/Reuters
Police officers stood guard in Berlin as Germans demonstrated against coronavirus measures on Wednesday.Credit…Christian Mang/Reuters
BERLIN — State lawmakers in Germany approved a new version of a law on Thursday boosting the federal government’s power to enforce uniform coronavirus lockdown rules. New restrictions are expected in most districts soon after the president signs the bill into law, which could be as early as Thursday afternoon.
The law, which Chancellor Angela Merkel’s cabinet passed last week, is a response to a disjointed virus response by state governments, which previously had the ultimate say in carrying out restrictions. For months, experts have called for a lockdown to control Germany’s surging third wave of coronavirus infections.
Under the law passed by the federal council of states on Thursday, the rules would apply uniformly across the country but would depend on the rate of infection in each district, leading to more severe lockdowns in highly affected areas. There would be a curfew from 10 p.m. to 5 a.m. in districts with more than 100 new infections per 100,000 people in a week. Restaurants would remain closed, and nonessential stores would require an appointment and a negative test result in districts with more than 150 new infections per 100,000 people. Schools would close if 165 new infections per 100,000 were registered.
Germany is currently measuring 161 infections per 100,000 in a week, according to the health authorities, which also counted 29,518 new infections on Wednesday.
As many as 8,000 people, including right-wing extremists and coronavirus deniers, took to the streets in Berlin to protest the measures on Wednesday. Several lawsuits against it have already been announced.
Germany has recorded more than 80,000 deaths so far.
In other developments across the world:
-
Japan’s auto industry group canceled the biennial Tokyo Motor Show, scheduled for the fall, because of rising coronavirus cases, the Kyodo News agency reported. It was the first cancellation in the 67-year history of the event, which drew around 1.3 million people in 2019. Akio Toyoda, the chairman of the industry group and president of Toyota Motor Corp., said at a news conference that “it seems difficult to offer main programs in a safe environment.” The cancellation came as Japan reported 5,291 new infections, the highest daily total in three months. And it raised more questions about plans for the Tokyo Olympics, which organizers have insisted will begin in July even as officials plan to impose emergency measures in Tokyo and other municipalities.
-
The European Union will not order an extra 100 million vaccines from AstraZeneca foreseen in its contract, a European Commission spokesman said Thursday, underscoring the soured relationship between the pharmaceutical company and the bloc of 27 countries. The bloc could have added 100 million doses of vaccines to its existing order of 300 million from AstraZeneca but the time to do so has passed, Stefan de Keersmaecker, the spokesman, said. The European Union is embroiled in a dispute with the British-Swedish company over its inability to deliver expected doses, which has set the bloc’s vaccination efforts back significantly. They have been in a legal arbitration process for weeks, and the bloc is considering suing.
 Megan Fairchild practicing in her parent’s home in Utah.Credit…Kim Raff for The New York Times
Megan Fairchild practicing in her parent’s home in Utah.Credit…Kim Raff for The New York Times
At the beginning of the pandemic, one of Megan Fairchild’s former dance teachers gave her some advice: Now would be a really great time to get pregnant. Ms. Fairchild, a principal at New York City Ballet, was aghast.
“I was like, that’s a ridiculous idea and the last thing on my mind right now,” she said. “This is going to last a couple months, and I don’t want to not be there when we get back.”
But when it became clear that her kind of live performance, dancing for thousands at Lincoln Center, would not be resuming anytime soon, the decision to have another child came to her in three words when she was meditating: Do it now.
For much of the pandemic year, Ms. Fairchild, 36, was pregnant with twins. On April 10, she gave birth to two girls.
She’s not the only one to have taken advantage of the theatrical shutdown. The dance world is experiencing a full-blown baby boom.
 Federal regulators have found many shortcomings at a plant of Emergent BioSolutions in Baltimore.Credit…Saul Loeb/Agence France-Presse — Getty Images
Federal regulators have found many shortcomings at a plant of Emergent BioSolutions in Baltimore.Credit…Saul Loeb/Agence France-Presse — Getty Images
WASHINGTON — Federal regulators have found serious flaws at the Baltimore plant that had to throw out up to 15 million possibly contaminated doses of Johnson & Johnson’s coronavirus vaccine, casting doubt on further production in the United States of a vaccine that the government once viewed as essential in fighting the pandemic.
The regulators for the Food and Drug Administration said that the company manufacturing the vaccine, Emergent BioSolutions, may have contaminated additional doses at the plant. They said the company failed to fully investigate the contamination, while also finding fault with the plant’s disinfection practices, size and design, handling of raw materials and training of workers.
The F.D.A. has not yet certified the plant, in Baltimore’s Bayview neighborhood, and no doses made there have gone to the public. All the Johnson & Johnson shots that have been administered in the United States have come from overseas.
The report amounted to a harsh rebuke of Emergent, which had long played down setbacks at the factory, and added to problems for Johnson & Johnson, whose vaccine had been seen as a game changer because it requires only one shot, can be produced in mass volume and is easily stored.
The inspection began after routine checks showed that Emergent workers had contaminated at least part of a batch of 13 million to 15 million doses of the Johnson & Johnson vaccine with the harmless virus that is used to make the AstraZeneca shot, which is not yet authorized in the United States.
The F.D.A. findings, based on an inspection that ended on Tuesday, underscore questions raised in reports by The New York Times about why Emergent did not fix problems earlier and why federal officials who oversee its lucrative contracts did not demand better performance.
In statements on Wednesday, the F.D.A., Emergent and Johnson & Johnson all said they were working to resolve the problems at the factory. There was no indication of how long that would take.
 Nepal’s dethroned king, Gyanendra Shah, center, at Golden Temple in Amritsar, India, last year.Credit…Sameer Sehgal/Hindustan Times, via Getty Images
Nepal’s dethroned king, Gyanendra Shah, center, at Golden Temple in Amritsar, India, last year.Credit…Sameer Sehgal/Hindustan Times, via Getty Images
KATHMANDU, Nepal — At the beginning of this month, Nepal’s dethroned king, Gyanendra Shah, and his wife, Komal, traveled to northern India for the Kumbh Mela, a Hindu pilgrimage where millions seek a dip in the Ganges River to absolve themselves of their sins.
Gyanendra bathed in the river, and for 10 days, he and his aides mingled in crowds and met ascetics, Hindu leaders and other dignitaries. On April 18, he and Komal flew home to Nepal, where supporters welcomed them at the airport and formed a procession to escort them home, chanting pro-Hindu and pro-monarchy slogans along the way.
Three days later, the couple tested positive for the coronavirus. Now they are in quarantine at their residence in Kathmandu, the capital, while health officials in Nepal try to trace anyone who was in contact with them.
“Both king and queen have isolated themselves from other family members,” said Phani Raj Pathak, an aide to Gyanendra, who was dethroned when Nepal became a republic in 2008 and ended a two-century-old Hindu monarchy. The former ruler, who is in his 70s, retains support among some Hindus in Nepal as well as among critics of the elected government.
The infections have cast a harsh spotlight on the Kumbh Mela, where millions of Hindu pilgrims have gathered for weeks, shoulder to shoulder and often maskless, even as highly infectious variants of the coronavirus surge across South Asia. On Thursday, India reported more than 312,000 new infections, the highest daily total in any country since the pandemic began.
The Indian government has defended the gathering as safe, even as news media report thousands of infections among participants. Organizers say that attendees are required to wear masks and show proof of a negative coronavirus test, but they acknowledge that given the size of the event, many could have flouted the rules.
Now there are fears that the Kumbh Mela will cause the virus to explode in Nepal, which shares a porous border with India.
“The majority of people weren’t wearing face masks,” said Yogini Saritanandi, a pilgrim who returned to Nepal. She said she had seen “nothing other than a sea of humans on the bank of the Ganges.”
She said the authorities in the northern city of Haridwar, where the Kumbh Mela is being observed this year, began to slightly restrict entry after a few ascetics were reportedly infected and after India’s prime minister, Narendra Modi, urged organizers to observe social distancing. But it appeared to be too late.
“People got Covid one after another,” said Ms. Saritanandi, 43. “When I saw this, I thought of my 10-year-old son, and I cut my visit short to return to Nepal earlier.”
As Indian states impose new lockdowns, tens of thousands of Nepali migrant workers have returned from India without undergoing coronavirus tests. After reporting no new infections for much of January, Nepal is now averaging more than 1,100 cases a day, according to a New York Times database.
The government has closed schools and colleges in urban areas and tried to speed up vaccinations, with more than 1.7 million people having received at least one shot. But the inoculation drive was slowed after India restricted exports of vaccines to fight the outbreak at home, leaving Nepal to rely on a donation of shots from China.
 A man used a self-administered coronavirus test kit in Durham, N.C., in February.Credit…Pete Kiehart for The New York Times
A man used a self-administered coronavirus test kit in Durham, N.C., in February.Credit…Pete Kiehart for The New York Times
The health effects of Covid-19 not only can stretch for months, but also appear to increase the risk of death and chronic medical conditions even in people who were never sick enough with Covid to be hospitalized, according to a new study published Thursday in the journal Nature.
Researchers looked at medical records of more than 73,000 people across the United States who were infected with the coronavirus between March and November 2020 and did not require hospitalization. In the period from one to six months after becoming infected, those patients were 20 percent more likely to need outpatient medical care, and 60 percent more likely to die, than people who had not contracted the coronavirus.
The Covid survivors experienced a vast array of long-term medical problems that they had never had before — not just lung issues from the respiratory effects of the virus, but symptoms that could affect virtually any organ system or part of the body, from neurological to cardiovascular to gastrointestinal. They were also at greater risk of mental health problems, including anxiety and sleep disorders.
Some of the patients’ post-Covid medical issues — like diabetes, kidney disease and some heart problems — could become chronic conditions that would require treatment for the rest of their lives.
Most of the nearly 32 million people who have contracted the coronavirus in the United States have not needed hospitalization, so the findings may have wide implications. But the study sample and the control group they were compared with may not be very representative of the general public: They were Veterans Health System patients, overwhelmingly men with a median age over 60.
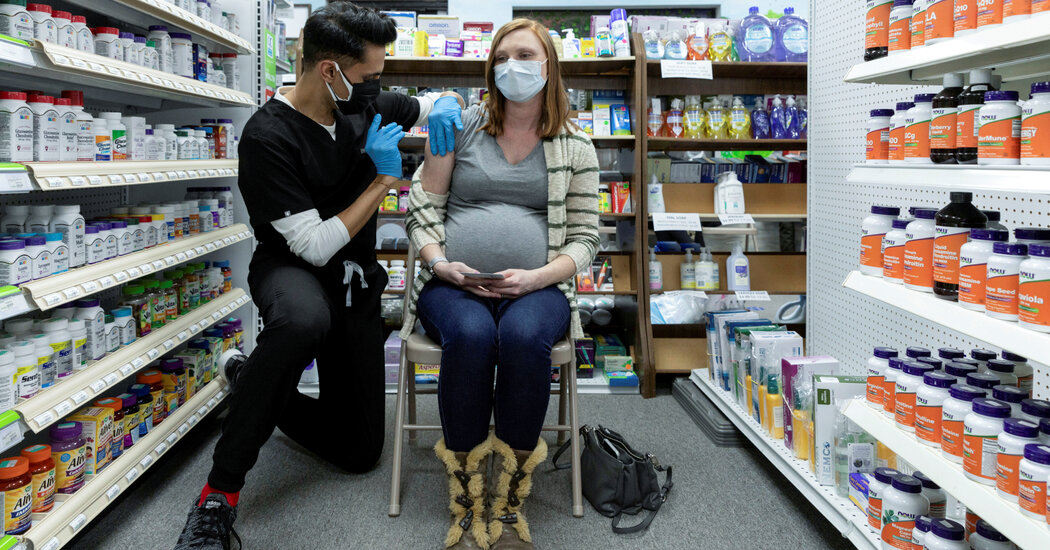
 A pregnant woman receiving the Pfizer vaccine in Schwenksville, Pa., in February.Credit…Hannah Beier/Reuters
A pregnant woman receiving the Pfizer vaccine in Schwenksville, Pa., in February.Credit…Hannah Beier/Reuters
In an early analysis of coronavirus vaccine safety data, researchers at the Centers for Disease Control and Prevention have found no evidence that the Pfizer-BioNTech or Moderna vaccines pose serious risks during pregnancy.
The findings are preliminary and cover just the first 11 weeks of the U.S. vaccination program. But the study, which included self-reported data on more than 35,000 people who received one of the vaccines during or shortly before pregnancy, is the largest yet on the safety of the coronavirus vaccines in pregnant people.
During the clinical trials of the vaccines, pregnant women were excluded. That left patients, doctors and experts unsure whether the shots were safe to administer during pregnancy.
“There’s a lot of anxiety about whether it’s safe and whether it would work and what to expect as far as side effects,” said Dr. Stephanie Gaw, a maternal-fetal medicine specialist at the University of California, San Francisco.
The new data, Dr. Gaw said, demonstrate that “a lot of pregnant people are getting the vaccine, there isn’t a significant increase in adverse pregnancy effects at this point, and that side effect profiles are very similar to nonpregnant people.”
“I think that’s all very reassuring,” she said, “and I think it will really help providers and public health officials more strongly recommend getting the vaccine in pregnancy.”
Covid-19 poses serious risks during pregnancy. Pregnant women who develop symptoms of the disease are more likely to become seriously ill, and more likely to die, than nonpregnant women with symptoms.
Because of those risks, the C.D.C. has recommended that coronavirus vaccines be made available to pregnant women, though it also suggests that they consult with their doctors when making a decision about vaccination.
The new study, which was published on Wednesday in The New England Journal of Medicine, is based largely on self-reported data from V-safe, the C.D.C.’s coronavirus vaccine safety monitoring system. Participants in the program use a smartphone app to complete regular surveys about their health, and any side effects they might be experiencing, after receiving a Covid-19 vaccine.
The researchers analyzed the side effects reported by V-safe participants who received either the Pfizer or Moderna vaccine between Dec. 14, 2020, and Feb. 28, 2021. They focused on 35,691 participants who said that they had been pregnant when they received the vaccine or became pregnant shortly thereafter.
After vaccination, pregnant participants reported the same general pattern of side effects that nonpregnant ones did, the researchers found: pain at the injection site, fatigue, headaches and muscle pain.
Women who were pregnant were slightly more likely to report injection site pain than women who were not, but less likely to report the other side effects. They were also slightly more likely to report nausea or vomiting after the second dose.
Pregnant V-safe participants were also given an opportunity to enroll in a special registry that tracked pregnancy and infant outcomes.
By the end of February, 827 of those enrolled in the pregnancy registry had completed their pregnancies, 86 percent of which resulted in a live birth. Rates of miscarriage, prematurity, low birth weight and birth defects were consistent with those reported in pregnant women before the pandemic, the researchers report.
“This study is of critical importance to pregnant individuals,” Dr. Michal Elovitz, a maternal-fetal medicine specialist at the University of Pennsylvania, said in an email. “It is very reassuring that there were no reported acute events in pregnant individuals” over the course of the study, she said.
But the report has several limitations and much more research is needed, experts said. Enrollment in the surveillance programs is voluntary and the data are self-reported.
In addition, because the study period encompassed just the first few months of the U.S. vaccination campaign, the vast majority of those enrolled in the pregnancy registry were health care workers. And there is not yet any data on pregnancy outcomes from people who were vaccinated during the first trimester of pregnancy.
“I think we can feel more confident about recommending the vaccine in pregnancy, and especially with pregnant people that are at risk of Covid,” Dr. Gaw said. “But we do need to wait for more data for complete pregnancy outcomes from vaccines early in pregnancy.”
 Jackie Robinson Day at Dodger Stadium earlier this month.Credit…Kirby Lee/USA Today Sports, via Reuters
Jackie Robinson Day at Dodger Stadium earlier this month.Credit…Kirby Lee/USA Today Sports, via Reuters
Fully vaccinated baseball fans will be granted their own section at the Los Angeles Dodgers game this weekend against the San Diego Padres.
The set-aside seats, reported by The Los Angeles Times, are part of the many incentives being offered — from doughnuts to beer — to encourage people to get vaccinated against Covid-19. The Miami Heat and the San Francisco Giants have introduced similar sections at their stadiums.
To prove they are fully vaccinated, fans will have to show government-issued I.D. and documentation like a vaccination card, according to the Dodgers’ website. Everyone 16 years and older will have to show proof that at least two weeks have passed since they were fully vaccinated. Fans younger than 16 will be required to show proof of a negative coronavirus test taken within 72 hours before admission.
Face masks will still be required, but social distancing will not. The team said spectators in the sections for the fully vaccinated will be seated directly next to each other.
The game Saturday won’t mark the first time fans have entered Dodger Stadium since the pandemic began. The team’s home opener on April 9 was attended by fans — just not all that many of them. Attendance was capped at around 11,000, about 20 percent of capacity.
In the past week, there has been an average of more than 2,300 daily coronavirus cases in the state, and Los Angeles County has seen an average of 435 daily cases — a 20 percent drop over the past two weeks, according to a New York Times database.
As of Wednesday, more than 40 percent of Californians had received at least one dose of the vaccine, and more than 20 percent had been fully vaccinated.
On April 15, Gov. Gavin Newsom loosened some restrictions in the state, permitting limited outdoor gatherings and live events, depending on a region’s Covid-19 risk level.
 A 5K run organized by New York Road Runners in October.Credit…John Minchillo/Associated Press
A 5K run organized by New York Road Runners in October.Credit…John Minchillo/Associated Press
New York Road Runners, the club that puts on the New York City Marathon, has announced the return of its first regularly scheduled race since the beginning of the pandemic.
On Thursday, the club said that it would hold the annual New York Mini 10K on June 12. The 10-kilometer, women-only race has been held annually since 1972, with the exception of last year.
“This is our first real table setting,” said Kerin Hempel, the organization’s interim chief executive. “It’s starting to feel like ‘OK, we’re back, we’re coming back.’”
This will not be the first race the club has held since the onset of the pandemic.
The organization has held a series of “return to racing” events as pilots starting last fall, allowing very small fields to run with safety protocols in place. Among other measures, the races had temperature checks, staggered starts and different corralling of runners.
Those events, Ms. Hempel said, have given N.Y.R.R. the confidence to move ahead with its first regularly scheduled race since March 2020.
The Mini 10K field will be smaller than in past years, with a cap of 1,200 runners. The race will also have safety protocols, such as requiring runners to mask up at the start and finish. (They will be strongly encouraged to wear masks during the race, too.)
It will be the first time N.Y.R.R. has welcomed elite athletes since the 2019 New York City Marathon, with 25 elite athletes expected at the starting line. The 2019 Mini 10K champion, Sara Hall, will return to defend her title.
The announcement comes as runners look ahead — with cautious optimism — to the return of major road races. Ms. Hempel anticipated the question on the minds of many: What does this mean for the New York City Marathon?
“We’ve been saying the marathon is going to happen,” she said. “It’s more about what it’s going to look like, and how many people we can accommodate on the course.”