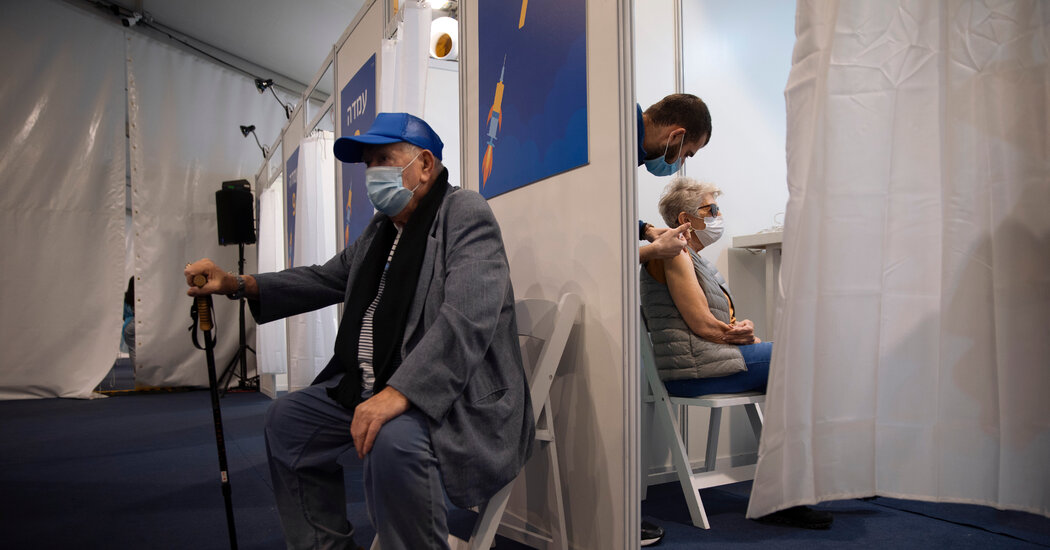
A health care worker administers a Covid-19 vaccine at Clalit Health Services in the ultra-Orthodox Israeli city of Bnei Brak on January 6, 2021.
JACK GUEZ | AFP | Getty Images
As the US, UK and Europe try to speed up their own Covid vaccination campaigns, one country is surpassing them all: Israel.
Israel’s vaccination campaign began on December 19 with Prime Minister Benjamin Netanyahu, the first person to be vaccinated in the country. Priority will be given to people over 60, healthcare workers and all clinically vulnerable people – reportedly making up around a quarter of the 9 million population.
It is ahead of other countries that have also started introducing vaccinations. To date, experts have said that around 1.5 million people in Israel received their first vaccine shot as a new lockdown came amid an increase in coronavirus cases.
According to Dr. Boaz Lev, chairman of the Disease Control and Coronavirus Vaccines Advisory Committee, has now vaccinated around 60% of the priority groups for the vaccine, although some of them are difficult to reach, such as those who only live at home by Israel’s Ministry of Health. The country is vaccinating around 150,000 people daily, he added, and intends to have vaccinated most of the country by April.
“The main goal of our vaccination program is to vaccinate as many people as possible as quickly as possible,” said Lev.
Lessons for the rest of the world
From logistics to public information campaigns, there are a number of lessons other countries could learn from trying to speed up their own vaccination campaigns.
“First of all … plan ahead. Be prepared, run a big information campaign and gain people’s trust, that’s on one side,” Lev told CNBC on Wednesday.
“Then you create a good flow of vaccines, a good flow of people … with a good administrative background so you can register them and let them know when to come for their next push. So there are a lot of things that which is basically about planning ahead and rolling it out to make it flow. “
In this aerial photo, taken in Tel Aviv, Israel, on Monday January 4, 2020, people are queuing outside a Covid-19 mass vaccination center in Rabin Sqaure. Israel plans to vaccinate 70% to 80% of its population by April or May. Health Minister Yuli Edelstein has said.
Bloomberg | Bloomberg | Getty Images
Israeli officials weren’t sure how many vaccinations the country ordered, but vaccine manufacturers reported that they received 8 million doses of the Pfizer / BioNTech vaccine and 6 million doses of the Moderna vaccine (the first batch of which was due to it) Arrival Thursday). It was not disclosed how much Oxford University / AstraZeneca vaccine the country ordered.
All of these vaccines require that each have two doses; There are reports that Israel paid higher vaccine prices than it competed to supply larger countries.
Lev said Israel’s ambitious goal of vaccinating the majority of its population through its public hospitals and vaccination centers requires careful planning. “We have to set up the logistics for this, and that takes a huge effort,” he said.
“The next is to be in the correct order in vaccinating people. Unless we have an abundance of vaccines … we need to have a very orderly queue so we know who is being vaccinated, and that should be loud some Principles, “he added. “It should be safe, it should be flexible, it should be as simple as possible, but it should also follow the principle that those who are more vulnerable should get it first … to avoid mortality and morbidity (of the pandemic) . “
Logistics and sales
Public health experts told CNBC that there were a number of factors that made it possible for Israel to vaccinate so efficiently, including the small population and geography and the efficiency of its health system.
Israel has a public health system in which everyone has to belong to one of four health organizations (HMOs) that work a bit like the UK’s National Health Service. Vaccine supplies were distributed to these HMOs, who in turn distributed them to their respective members.
Ronit Calderon-Margalit, professor of epidemiology at Hadassah-Hebrew University’s Braun School of Public Health, told CNBC on Wednesday that the vaccination campaign exceeded their expectations. “It’s amazing, it’s way beyond my wildest dreams and I don’t get to say that often,” she said.
People will receive a dose of the Pfizer-BioNTech Covid-19 vaccine at a Covid-19 mass vaccination center on Rabin Square in Tel Aviv, Israel on Monday January 4, 2020.
Bloomberg | Bloomberg | Getty Images
She attributed part of this success to the efficiency of the four HMOs: Clalit, Maccabi, Meuhedet and Leumit or “Kupot Cholim” as they are collectively known.
“They all have vaccines from the government to vaccinate the population, and they are very good at the logistics of distributing services that vaccines,” she said. Experts told CNBC that at the end of the day, hospitals and clinics are also giving the vaccines to people outside of the priority groups so as not to waste supplies.
The Israeli health system is heavily digitized, so anyone who receives the vaccine is registered as such by the Ministry of Health.
Israel recorded 466,916 cases of the virus and 3,527 deaths as of Thursday, according to Johns Hopkins University. As in other countries, there has been an increase in infections over the winter.
On Wednesday, Netanyahu blamed a new, more transmissible strain of virus, first identified in the UK (what he called the “British mutation”), responsible for an increase in cases in the country. Due to the wave of infections, Israel will enter a new strict lockdown for two weeks on Thursday at midnight.
In addition to vaccination centers and clinics, hospitals are of course at the forefront.
Yoel Har-Even is Director of International and Resource Development at Sheba Medical Center, the largest hospital in the Middle East (and by the way, where Netanyahu was vaccinated in December).
He told CNBC on Wednesday that his hospital had vaccinated around 45,000 people in the past two weeks.
These people range from the most at risk, including police officers and Holocaust survivors, an experience that Har-Even said was very moving, to teachers. He said everyone he met was happy to have received the vaccine (sentiment against vaccines is low in Israel) and the mainstream media of all political lines supported the vaccination campaign.
“We understand that this is a crucial time and everyone here agrees,” said Har-Even. “It reminds us a little of a time of war in Israel and when there is war there is unity.”
He added that people’s acceptance and willingness to receive the vaccine is a cause for great pride.
“You just have to see the lines and the queues of people standing still, there is no pushing or screaming,” he said. “The time of the corona means (the vaccination campaign) that it runs faster, quieter and with much, much more order and efficiency in the process.”