Here’s what you need to know:
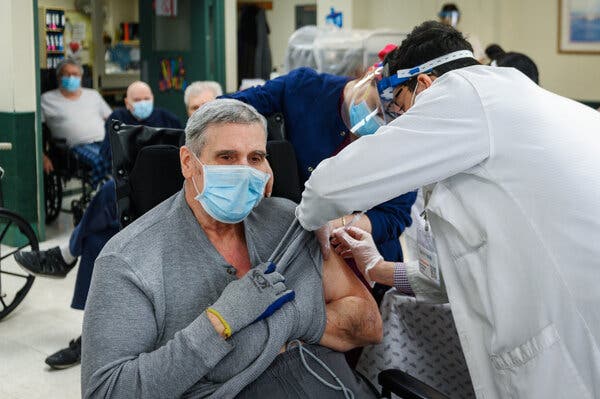
James Ivialiotis received his second dose of a Covid-19 vaccine at a nursing home on Staten Island, N.Y., on Monday.Credit…Christopher Occhicone for The New York Times
The Trump administration, in a major policy shift aimed at accelerating lagging distribution of the coronavirus vaccine, announced on Tuesday that it would release all available doses and instructed states to immediately begin vaccinating every American 65 and older, as well as tens of millions of adults with health conditions that put them at higher risk of dying from the virus.
The announcement, by Health Secretary Alex M. Azar II and other top federal health officials, came amid continuing complaints about the pace of the vaccine rollout. Mr. Azar warned that states will lose their allocations if they don’t use up doses quickly, and that starting in two weeks, how many each state receives will be based on the size of its population of people 65 and older.
Precisely how that will work is unclear; in two weeks, President-elect Joseph R. Biden Jr. will already have been sworn in as president. Mr. Azar said the incoming Biden administration would be briefed on the changes, though he added that Americans “operate with one government at a time, and this is the approach that we believe best fulfills the mission.”
The new distribution plan, first reported Tuesday morning by Axios, is a reversal for the administration, which had been holding back roughly half of its vaccine supply — millions of vials — to guarantee that second doses would be available. Mr. Azar said the administration always expected to make the shift when it was confident in the supply chain. Both vaccines authorized in the United States so far require two doses: 21 days apart for the one developed by Pfizer and BioNTech, and 28 days apart for the one from Moderna.
“This next phase reflects the urgency of the situation we face,” he said.
Just days ago, Mr. Azar and officials from Operation Warp Speed, the administration’s fast-track vaccine initiative, criticized aides to Mr. Biden for announcing a similar plan. Mr. Azar said at the time that releasing nearly all of the doses, as the Biden team proposed, would jeopardize the “system that manages the flow, to maximize the number of first doses, but knowing there will be a second dose available.”
He called any proposed changes an “untenable position.”
Health officials also recommend that the vaccines be given to all adults with pre-existing conditions that make them more likely to develop serious illness from the virus, such as diabetes, chronic lung or heart disease, high blood pressure and cancer. Before the change, the vaccines were largely being distributed to people in the highest-risk categories, including frontline health care workers and older people in nursing homes.
In addition to the eligibility changes, health officials are also adding more community centers and pharmacies to the list of places where people can be vaccinated.
Mr. Azar’s new directive threatens to create more confusion in states that had already articulated different plans for who should receive the vaccine next. As of Monday, about 9 million people have received at least one dose of a Covid-19 vaccine, according to the Centers for Disease Control and Prevention, far short of the federal government’s original goals. At least 151,000 people in the United States have been fully vaccinated, as of Jan. 8, according to a New York Times survey of all 50 states. More than 375,000 people have died related to the virus and in recent days, the number of daily deaths in the country has topped 4,000.
Instead of holding back vaccine doses all existing doses will be now sent to states to provide initial inoculations. Second doses are to be provided by new waves of manufacturing.
The idea of using existing vaccine supplies for first doses has raised objections from some health workers and researchers, who worry that frontloading shots will raise the risk that second injections will be delayed. Clinical studies testing the vaccines showed the shots were effective when administered in two-dose regimens on a strict schedule. And while some protection appears to kick in after the first shot, experts remain unsure of the extent of that protection, or how long it might last without the second dose to boost its effects.
But others have vocally advocated for explicit dose delays, arguing that more widely distributing the partial protection afforded by a single shot will save more lives in the meantime.
The new recommendations come after some states have already begun vaccinating people 65 and older, leading to long lines and confusion over how to get a shot. Health experts and officials have faced difficult choices as they decided which groups would be prioritized in the vaccine rollout. While the elderly have died of the virus at the highest rates, essential workers have borne the greatest risk of infection, and the category includes many poor people and people of color, who have suffered disproportionately high rates of infection and death.
Despite the bumpy rollout, Gov. Ron DeSantis of Florida, who prioritized people 65 and older from the start, said he believed making all older people eligible was always the right thing to do.
The initial guidelines “would have allowed a 20-year-old healthy worker to get a vaccine before a 74-year-old grandmother,” he said on Tuesday at a news conference in the sprawling retirement community of The Villages. “That does not recognize how this virus has affected elderly people.”
In New York, which began vaccinating people 75 and older and more essential workers this week, Gov. Andrew M. Cuomo said that the state will accept the new federal guidance to prioritize those 65 and older, though he criticized the administration for not clearly defining who should be considered “immunocompromised.”
The new guidance will make more than 7 million New Yorkers eligible for the vaccine, Mr. Cuomo said, though the state only receives 300,000 doses a week.
“The federal government didn’t give us an additional allocation,” he said. “At 300,000 per week, how do you effectively serve 7 million people, all of whom are now eligible, without any priority?”
New Yorkers 65 and older are immediately able to schedule appointments on the state’s website, according to Melissa DeRosa, a top Cuomo aide, who added that the state was working with the C.D.C. on who is considered immunocompromised.
New guidelines released on Monday by the Centers for Disease Control and Prevention now note that while people should get their second shots “as close to the recommended 3-week or 1-month interval as possible,” there is “no maximum interval between the first and second doses for either vaccine.”
The update perplexed experts, who said that while other, previously licensed vaccines that involve multiple doses can be administered months or even years apart, no evidence yet exists to clearly support this strategy for Covid-19. “They will need to back this up with data,” said Marion Pepper, an immunologist at the University of Washington.
Dr. Leana Wen, an emergency physician at the George Washington School of Public Health, echoed the call for an explanation. With skepticism of vaccines already hindering the rollout of some shots, “the last thing we want to do is give the impression that there are shortcuts being taken in the approval process.”
Health officials in Britain are now allowing intervals between the first and second doses of Pfizer’s vaccines of up to 12 weeks. Last week, the World Health Organization said the injections could be given up to six weeks apart. The agency’s Strategic Advisory Group of Experts on Immunization “considers the administration of both doses within 21 to 28 days to be necessary for optimal protection,” said Saad Omer, a vaccine expert at Yale University who helped draft the WHO’s position on the matter.
In response to queries about dose delays, representatives from Pfizer and Moderna have repeatedly pointed to the company’s clinical trials, which tested dosing regimens of two shots, separated by 21 days for Pfizer, and 28 days for Moderna.
“Two doses of the vaccine are required to provide the maximum protection against the disease, a vaccine efficacy of 95 percent,” Steven Danehy, a spokesman for Pfizer, said earlier this month. “There are no data to demonstrate that protection after the first dose is sustained after 21 days.”
| United States › |
United StatesOn Jan. 11 |
14-day change |
| New cases |
222,902 |
+37% |
|
| New deaths |
2,048 |
+48% |
|
| World › |
WorldOn Jan. 11 |
14-day change |
| New cases |
625,815 |
+32% |
|
| New deaths |
10,307 |
+28% |
|
Where cases per capita are
highest
 A coronavirus testing site in a shopping center parking lot in southern Los Angeles last week.Credit…Philip Cheung for The New York Times
A coronavirus testing site in a shopping center parking lot in southern Los Angeles last week.Credit…Philip Cheung for The New York Times
California is trying to speed up its vaccination efforts, which have lagged amid the state’s struggle with a weekslong deluge of coronavirus cases that has led to some of the most dire consequences in the country.
Emergency rooms have had to shut their doors to ambulances for hours at a time. Nearly one in 10 people has tested positive for the virus in Los Angeles County, the nation’s most populous. And a surge of hospitalizations has caused problems for the oxygen delivery and supply system used by medical facilities.
Over the past week, an average of 480 people daily have died of Covid-19 in the state, according to a New York Times database.
Gov. Gavin Newsom said on Monday that California would employ an “all-hands-on-deck approach” to ramp up vaccinations.
The approach includes transforming Dodger Stadium from one of the nation’s biggest and most visible Covid-19 testing sites into a mass vaccination center. Petco Park, where the San Diego Padres play, and the state fairgrounds in Sacramento are also being set up as vaccination sites, the governor said.
The Orange County board of supervisors said on Monday that the county’s first of five planned “super” vaccination sites would open this week at the Disneyland Resort in Anaheim, which has been closed for much of the pandemic. Vaccinations will be available by appointment to everyone in “Phase 1a,” which includes frontline health care workers, paramedics, dentists and pharmacists.
Los Angeles County opened vaccine eligibility to a wider group of health care workers on Monday, allowing workers in facilities like primary care clinics, Covid-19 testing centers, laboratories, pharmacies and dental offices, as well as those who work with people who are homeless, to be vaccinated.
Previously, workers in hospitals and long-term-care facilities were prioritized. But as The Los Angeles Times reported, large numbers of health care workers in Los Angeles and Riverside Counties were declining to be inoculated.
And relatively few people in California have gotten vaccine doses, compared with other places: Only 2 percent of the state’s population has received a vaccine, according to a New York Times database; 782,638 doses out of the more than 2.8 million that the state has received have been administered.
Video
transcript
Back
transcript
Newsom Broadens Who Can Administer Vaccines
Gov. Gavin Newsom of California described an “all-hands-on-deck approach” that will allow a wider range of health care workers, including pharmacists and dentists, to administer the coronavirus vaccine.
We are sending an urgent call across the spectrum, our health care partners, our legislative partners, as well as labor and business partners up and down the state, this notion of an all-hands-on-deck approach to accelerate the equitable and safe distribution of vaccines. Again, we’re not losing sight of the issue of equity. We’re not losing sight of the imperative to prioritize the most vulnerable and the most essential. So that’s why we talk about our special efforts to vaccinate the vaccinators as part of an all hands on deck — the slide that represents the number of categories of individuals and groups that can currently vaccinate. And you can see the myriad of different registered nurses, physician assistants and the like. But we recognize more folks need to have that ability. And that’s why you recall a week or so ago, we talked about our efforts on pharmacists and pharm techs. We’re seeing more and more paramedics partnering with the counties. Local health officers are encouraging this and we are very supportive of EMTs as this local option for additional vaccinators to help administer these vaccines faster.
 Gov. Gavin Newsom of California described an “all-hands-on-deck approach” that will allow a wider range of health care workers, including pharmacists and dentists, to administer the coronavirus vaccine.CreditCredit…Alex Welsh for The New York Times
Gov. Gavin Newsom of California described an “all-hands-on-deck approach” that will allow a wider range of health care workers, including pharmacists and dentists, to administer the coronavirus vaccine.CreditCredit…Alex Welsh for The New York Times
Dr. Mark Ghaly, California’s secretary of health and human services, said at a news conference on Monday that the state was working to distribute vaccines to those who need them and want them — without allowing wealthy people to cut the line.
Mr. Newsom said the state was allowing a broader range of workers to administer vaccines, including pharmacists and dentists, and was rolling out a public awareness campaign in 18 languages.
“People have said, ‘Well, what about sending in the National Guard?’” he said of the groups administering vaccines. “Well, we have the National Guard out there.”
He also said there were urgent efforts to “vaccinate the vaccinators.”
 Representative Brad Schneider, Democrat of Illinois, speaking in Washington last year.Credit…Samuel Corum/Getty Images
Representative Brad Schneider, Democrat of Illinois, speaking in Washington last year.Credit…Samuel Corum/Getty Images
Three Democratic members of Congress have tested positive for the coronavirus, and say they believe their infections are linked to their time spent in a secure location with colleagues who did not wear masks during last week’s siege of the U.S. Capitol.
Representative Brad Schneider, Democrat of Illinois, said he received a positive test result Tuesday morning after driving home to Illinois, and that he did not have symptoms. Like Representatives Bonnie Watson Coleman of New Jersey and Pramila Jayapal of Washington, two Democrats who had announced positive tests on Monday, he directly blamed a group of House Republicans who refused to wear masks while sheltering in a secure location during the Capitol siege.
“Today, I am now in strict isolation, worried that I have risked my wife’s health and angry at the selfishness and arrogance of the anti-maskers who put their own contempt and disregard for decency ahead of the health and safety of their colleagues and our staff,” Mr. Schneider said.
He called for lawmakers who ignore public health guidance to be sanctioned “and immediately removed from the House floor by the Sergeant-at-arms for their reckless endangerment of their colleagues.”
Capitol Hill has long struggled to contain the spread of the virus, and within hours of the beginning of the 117th Congress on Jan. 3, lawmakers began announcing positive test results.
Now lawmakers, aides, police officers and reporters who fled to secure locations during the siege have been warned that they might have been exposed to the virus while sheltering from the mob.
On Sunday, Representative Chuck Fleischmann, Republican of Tennessee, who was also in protective isolation at the Capitol during the siege, said that he had tested positive for the virus after being exposed to his roommate, Representative Gus Bilirakis of Florida, also a Republican.
Mr. Fleischmann told the local news station WRCB that he was notified Wednesday that Mr. Bilirakis had tested positive, but did not receive the notification amid the riot. He said he did not know how many other lawmakers he had come in contact with.
Democrats, already frustrated by resistance from their Republican colleagues to wearing masks, accused maskless Republicans in the secure House location of reckless indifference.
“It angers me when they refuse to adhere to the directions about keeping their masks on,” Ms. Watson Coleman said in an interview. “It comes off to me as arrogance and defiance. And you can be both, but not at the expense of someone else.”
Ms. Jayapal said on Twitter that she had tested positive “after being locked down in a secured room at the Capitol where several Republicans not only cruelly refused to wear a mask but recklessly mocked colleagues and staff who offered them one.”
Ms. Jayapal, who said she had begun quarantining immediately after the siege on the Capitol, also said that any member of Congress who did not wear a mask should be removed from the floor by the sergeant-at-arms and fined.
“This is not a joke,” she said in a statement. “Our lives and our livelihoods are at risk, and anyone who refuses to wear a mask should be fully held accountable for endangering our lives because of their selfish idiocy.”
 Dustin Johnson teeing off the 17th tee during round two at the Masters golf tournament in Augusta, Ga., in November.Credit…Doug Mills/The New York Times
Dustin Johnson teeing off the 17th tee during round two at the Masters golf tournament in Augusta, Ga., in November.Credit…Doug Mills/The New York Times
This year’s Masters tournament in April will be attended by a limited number of spectators, the Augusta National Golf Club announced Tuesday. The club, which prohibited fans from the event two months ago, did not specify how many fans would be allowed in 2021, adding that spectators would be permitted if “it can be done safely.”
The 2020 Masters was postponed from its usual April date to November because of the coronavirus pandemic and was contested with protocols that included virus testing before the event for all players, caddies, club members, staff and other personnel, including a reduced number of media members.
Fred Ridley, the club chairman, said in a statement issued Tuesday that similar health standards would be instituted for this year’s tournament, which is scheduled to be contested from April 8 to 11. The club, based in Augusta, Ga., made the announcement as the state reported 16 new coronavirus deaths and 7,957 new cases on Jan. 11. Over the past week, there has been an average of 9,604 cases per day, an increase of 55 percent from the average two weeks earlier.
“Following the successful conduct of the Masters Tournament last November with only essential personnel, we are confident in our ability to responsibly invite a limited number of patrons to Augusta National in April,” Ridley said. “As with the November Masters, we will implement practices and policies that will protect the health and safety of everyone in attendance.”
The Augusta National statement said the club was in the process of communicating with all ticket holders and that refunds will be issued to those patrons not selected to attend.
 Commuters at Shinjuku station in Tokyo last week.Credit…Noriko Hayashi for The New York Times
Commuters at Shinjuku station in Tokyo last week.Credit…Noriko Hayashi for The New York Times
Another new coronavirus variant has been detected in four people who traveled to Japan from Brazil.
Japan’s health ministry said that the people who arrived this month at Tokyo’s Haneda Airport had tested positive for the coronavirus and that it was a separate variant with similarities to those detected in Britain and South Africa. It is also distinct from another variant recently identified in Brazil, according to experts who have analyzed the data.
Makoto Shimoaraiso, an official with Japan’s Cabinet Secretariat and Office for Covid-19 Preparedness and Response, said on Tuesday that the country was consulting with the World Health Organization.
It is not unusual for viruses to accumulate mutations or for new variants to emerge. But scientists are calling for greater surveillance of variants, particularly after those from Britain and South Africa proved to be more contagious.
Mr. Shimoaraiso said epidemiologists were not sure whether the variant identified in Japan was more infectious or likely to cause more severe illness.
According to Japan’s health ministry, one of the passengers infected with the new variant, a man in his 40s, was admitted to a hospital after having breathing difficulties. Of the other cases, a woman in her 30s and a teenage boy are experiencing sore throats and fever, and a teenage girl is asymptomatic.
 London last week. A coronavirus variant that emerged in Britain has been found in about 50 countries.Credit…Andrew Testa for The New York Times
London last week. A coronavirus variant that emerged in Britain has been found in about 50 countries.Credit…Andrew Testa for The New York Times
In recent weeks, scientists have raised concerns about a coronavirus variant first detected in December in South Africa, noting that this version of the virus may spread more quickly than its cousins, and perhaps be harder to quash with current vaccines.
Their worries are compounded by skyrocketing Covid-19 cases in the United States and another highly infectious new variant that is driving a surge in Britain.
Scientists still have a lot to learn about these variants, but experts are concerned enough to warn people to be extra-vigilant in masking and social distancing. Here’s what you need to know:
-
The British variant has been found in about 50 countries, including the United States, where dozens of cases have been identified. The South African variant has spread to about 10 countries but has yet to be detected in the United States.
-
Both variants carry genetic changes in the virus’s spike protein — the molecule used to unlock and enter human cells — that could make it easier to establish an infection. Researchers estimate that the British variant is about 50 percent more transmissible than its predecessors. Julian Tang, a virologist at the University of Leicester, said that researchers didn’t yet have a good estimate for how much more contagious the South African variant is.
-
There is no evidence that any of the new variants are more deadly on their own, but an uptick in the spread of any virus creates ripple effects as more people become infected and ill. That can strain already overstretched health care systems and undoubtedly lead to more deaths.
-
It is unlikely that either variant will completely evade the protective effects of the new Covid vaccines. A recent study, not yet published in a scientific journal, found that the Pfizer-BioNTech vaccine is still effective against a virus carrying a mutation common to both new variants.
The South African variant does carry genetic changes that could make vaccines less effective: One mutation appears to make it harder for antibodies produced by the immune system to recognize the coronavirus, which means they may be less effective at stopping the variant. But it is “important to note that doesn’t mean vaccines won’t be functionally protective,” said Angela Rasmussen, a virologist affiliated with Georgetown University.
Vaccines use multifaceted immune responses, and while some antibodies may be confused by the variant, others probably won’t be. In addition, antibodies are only one sliver of the complex cavalry of immune cells and molecules that battle infectious invaders.
Also, if the virus accumulates more genetic changes, many of the authorized vaccines, including Pfizer’s and Moderna’s, can be adjusted fairly quickly.
 Transportation emissions dropped sharply in 2020 as millions of people stopped driving to work and lockdowns were in place.Credit…Lucy Nicholson/Reuters
Transportation emissions dropped sharply in 2020 as millions of people stopped driving to work and lockdowns were in place.Credit…Lucy Nicholson/Reuters
America’s greenhouse gas emissions from energy and industry plummeted more than 10 percent last year, reaching their lowest levels in at least three decades as the pandemic slammed the brakes on the nation’s economy, according to an estimate published Tuesday by the Rhodium Group.
The steep drop was the result of extraordinary circumstances, however, and experts say the United States still faces enormous challenges in getting its planet-warming pollution under control.
“The most significant reductions last year were around transportation, which remains heavily dependent on fossil fuels,” said Kate Larsen, a director at Rhodium Group, a research and consulting firm. “But as vaccines become more prevalent, and depending on how quickly people feel comfortable enough to drive and fly again, we’d expect emissions to rebound unless there are major policy changes put in place.”
Transportation, the nation’s largest source of greenhouse gases, saw a 14.7 percent decline in emissions in 2020 as millions of people stopped driving to work and airlines canceled flights. Although travel started picking up again in the second half of the year as states relaxed lockdowns, Americans drove 15 percent fewer miles last year than in 2019.
Over all, the fall in emissions nationwide was the largest one-year decline since at least World War II, the Rhodium Group said. It put the United States within striking distance of one of the major goals of the Paris climate agreement, a global pact by nearly 200 governments to address climate change.
As part of that agreement, President Barack Obama had pledged that U.S. emissions would fall 17 percent below 2005 levels by last year. President Trump withdrew the country from the Paris accord, and before last year, it appeared that the United States would miss the emissions target. But America’s industrial emissions are now roughly 21.5 percent below 2005 levels.
Scientists say that even a big one-year drop is not enough to stop climate change. Until humanity’s emissions are essentially zeroed out and nations are no longer adding greenhouse gases to the atmosphere, the planet will continue to heat up. As if to underscore that warning, European researchers announced last week that 2020 was probably tied with 2016 as the hottest year on record.
Global roundup
 Coronavirus testing at a clinic outside Kuala Lumpur, Malaysia, on Monday.Credit…Fazry Ismail/EPA, via Shutterstock
Coronavirus testing at a clinic outside Kuala Lumpur, Malaysia, on Monday.Credit…Fazry Ismail/EPA, via Shutterstock
Malaysia’s king declared a national state of emergency on Tuesday to stem a surge in coronavirus cases, suspending Parliament, closing nonessential businesses and locking down several states and territories, including the largest city, Kuala Lumpur.
The emergency declaration could last until Aug. 1, and some critics said the main beneficiary would be the prime minister, Muhyiddin Yassin, the head of an unelected government who for months has barely maintained his hold on power.
Mr. Muhyiddin, who asked the king to issue the declaration, went on television to assert that the emergency measure was necessary to contain the virus — and that it was not about extending his political career.
“Let me assure you, the civilian government will continue to function,” he said. “The emergency proclaimed by the king is not a military coup.”
Mr. Muhyiddin promised to hold a general election after the virus was brought under control.
Malaysia was mostly successful in containing the virus for much of last year, but the number of infections began rising in October and reached a daily peak of more than 3,000 new cases on Thursday. The surge was caused in part by an election campaign in the state of Sabah and by an outbreak among migrant workers. The government reported a total of more than 141,000 cases and 559 deaths as of Tuesday.
Mr. Muhyiddin came to power in March after the previous government collapsed. He formed a new coalition and the king appointed him prime minister without a parliamentary vote. Opponents have since questioned whether he has the support of a majority of Parliament’s 222 members.
Now, the king’s declaration means that no parliamentary vote or general election can be held for more than six months, as long as the virus persists.
James Chin, professor of Asian studies at the University of Tasmania, said the declaration gave Mr. Muhyiddin extraordinary powers, including the authority to pass laws that override existing ones and to use the military for police work.
“Politically he will benefit the most from this Covid emergency,” he said. “This will give him what he wants without any scrutiny from Parliament.”
Other global developments:
-
Taiwan on Tuesday reported two locally transmitted coronavirus infections: a doctor and a nurse at a hospital in the northern part of the island that treats coronavirus patients. They are Taiwan’s first locally transmitted cases since Dec. 22, when it reported the first such case since April.
-
The European Union’s top drug regulator said it would assess the coronavirus vaccine developed by AstraZeneca and Oxford University “under an accelerated timeline,” after receiving an application for emergency authorization of the drug.
-
The leader of the German state of Bavaria has urged health care workers to do their “civic duty” by getting vaccinated, and called on the government to consider making coronavirus vaccinations for medical personnel mandatory in some cases. And about half of the staff at Charité, Germany’s largest research hospital, has refused to receive vaccine shots, according to Dr. Andrej Trampuz, a department head at the facility.
-
Because of high infection numbers, Berlin residents will be restricted from traveling more than about 9 miles outside the city, under new rules agreed to by German lawmakers. The distance of travel within Berlin is not being limited.
-
A couple who were out walking on Saturday night in Sherbrooke, Quebec, told the police that they were in compliance with a new overnight curfew because the wife was walking her crawling husband on a leash like a dog, CTV News reported. People walking their dogs are excluded from the province’s curfew, which is in effect from 8 p.m. to 5 a.m., as are essential workers and those seeking medical care. The pair were fined 1,500 Canadian dollars each. The province’s leader, François Legault, said on Monday that 740 people were fined over the weekend for violating the curfew, the first of its kind in Canada.
 Dr. Rochelle P. Walensky is President-elect Joseph R. Biden Jr.’s nominee to lead the Centers for Disease Control and Prevention.Credit…Hilary Swift for The New York Times
Dr. Rochelle P. Walensky is President-elect Joseph R. Biden Jr.’s nominee to lead the Centers for Disease Control and Prevention.Credit…Hilary Swift for The New York Times
Dr. Rochelle P. Walensky, chief of the infectious diseases division at Massachusetts General Hospital and a professor at Harvard, has been nominated by President-elect Joseph R. Biden Jr. to be director of the Centers for Disease Control and Prevention. In a column for The New York Times Opinion section, excerpted here, she writes about her plans for the agency.
On Jan. 20, I will begin leading the C.D.C., which was founded in 1946 to meet precisely the kinds of challenges posed by this pandemic. I agreed to serve as C.D.C. director because I believe in the agency’s mission and commitment to knowledge, statistics and guidance. I will do so by leading with facts, science and integrity — and being accountable for them, as the C.D.C. has done since its founding 75 years ago.
I acknowledge that our team of scientists will have to work very hard to restore public trust in the C.D.C., at home and abroad, because it has been undermined over the last year. In that time, numerous reports stated that White House officials interfered with official guidance issued by the C.D.C.
As chief of the infectious diseases division at Massachusetts General Hospital, I and many others found these reports to be extremely disturbing. The C.D.C.’s science — the gold standard for the nation’s public health — has been tarnished. Hospitals, doctors, state health officials and others rely on the guidance of the C.D.C., not just for Covid-19 policies around quarantine, isolation, testing and vaccination, but also for staying healthy while traveling, strategies to prevent obesity, information on food safety and more.
Restoring the public’s trust in the C.D.C. is crucial. Hospitals and health care providers are beyond tired, beyond stretched. I know because I have stood among them, on the front lines of the Covid-19 response in Massachusetts. We also face the need for the largest public health operation in a century, vaccinating the population — twice — to protect ourselves and each other from a surging pandemic. Because the impact of Covid-19 does not fall equally on everyone, we must redouble our efforts to reach every corner of the U.S. population.
The research and guidance provided by the civil servants at the C.D.C. should continue regardless of what political party is in power. Novel scientific breakthroughs do not follow four-year terms. As I start my new duties, I will tell the president, Congress and the public what we know when we know it, and I will do so even when the news is bleak, or when the information may not be what those in the administration want to hear.
Video
transcript
Back
transcript
Gorillas Test Positive for Coronavirus at San Diego Zoo
Officials at the zoo’s Safari Park said that several gorillas had tested positive for the virus and that they believed an asymptomatic staff member infected the animals.
They’re doing OK, they’re experiencing some mild symptoms. And we continue to observe them. But they’re drinking, they’re eating and they’re interacting with one another. So we suspect that the gorillas got this virus from an asymptomatic team member. And that’s despite all of the precautions that we take. We follow C.D.C. guidelines. We follow San Diego County health guidelines. The team wears P.P.E. around all of our wildlife. And so even with all those precautions, we still have an exposure that we think happened with that team member. This virus has been very, very tricky. We’ve done everything we can to respond to it and make sure that we’re taking all the precautions and following all the guidelines that we can. But as we see it evolving everywhere around the world right now, we know that it is, it is, it’s evolving. It’s changing. And the best that we can do for humans and wildlife is just to ensure that we stay up to date on any protocols, that we remain nimble so that we can respond accordingly and make sure that we’re doing the very best we can to protect both our team, our guests and wildlife.
 Officials at the zoo’s Safari Park said that several gorillas had tested positive for the virus and that they believed an asymptomatic staff member infected the animals.CreditCredit…Ken Bohn/San Diego Zoo Global/Via Reuters
Officials at the zoo’s Safari Park said that several gorillas had tested positive for the virus and that they believed an asymptomatic staff member infected the animals.CreditCredit…Ken Bohn/San Diego Zoo Global/Via Reuters
Several gorillas at the San Diego Zoo Safari Park have tested positive for the coronavirus, becoming what federal officials say are the first known apes in the United States to be infected.
Zoo officials said on Monday that they believed the gorillas were infected by an asymptomatic staff member who had been following safety recommendations, including wearing personal protective equipment when near animals.
Veterinarians are closely monitoring the troop, which is made up of eight western lowland gorillas. The infected animals are expected to make a full recovery, officials said.
“Aside from some congestion and coughing, the gorillas are doing well,” Lisa Peterson, the Safari Park’s executive director, said in a statement.
Three animals are exhibiting symptoms, officials said. And because gorillas live together in troops, “we have to assume,” the zoo said, “that all members of the family group have been exposed.”
The total number of western lowland gorillas, which can be found in central Africa, has declined more than 60 percent over the past two decades, according to the World Wildlife Fund.
Zoo officials learned that at least two gorillas had been infected with the coronavirus after the animals were observed on Wednesday “coughing and showing other mild symptoms,” the zoo said in the statement.
The zoo’s Safari Park has been closed since Dec. 6 amid a lockdown, and the primate habitat where the gorillas are housed poses “no public health risk,” officials said. Last year, as the pandemic spread across the country, the zoo installed additional barriers to ensure that more than six feet of space separated visitors from “susceptible species,” officials said.
The gorillas are among the latest animals in the country to become infected with the coronavirus. In April, the first case of human-to-cat transmission was detected in a tiger at the Bronx Zoo in New York City. In August, minks on two farms in Utah tested positive. In December, a coronavirus infection in a snow leopard was detected at the Louisville Zoo in Kentucky.
Video Mayor Bill de Blasio of New York City announced on Tuesday that CitiField, the Mets’ home stadium in Queens, will be a “24/7 mega-vaccination site” starting the week of Jan. 25.CreditCredit…Ryan Christopher Jones for The New York Times
Mayor Bill de Blasio of New York City announced on Tuesday that CitiField, the Mets’ home stadium in Queens, will be a “24/7 mega-vaccination site” starting the week of Jan. 25.CreditCredit…Ryan Christopher Jones for The New York Times
Mayor Bill de Blasio of New York City announced on Tuesday that CitiField, the Mets’ home stadium in Queens, will be a mass vaccination site starting the week of Jan. 25. The site will operate around the clock, seven days a week, with the capacity to vaccinate 5,000 to 7,000 people a day, Mr. de Blasio said. The location is ideal, the mayor said, because it is right next to a subway and railroad station and has plenty of parking.
“It’s going to be big, and it’s going to be a game changer,” Mr. de Blasio said at a news conference on Tuesday.
Large sports venues across the country have been used as sites for mass coronavirus testing, and more recently for vaccination, including the home stadiums of the Los Angeles Dodgers and San Diego Padres baseball teams, the Arizona Cardinals of the N.F.L. and the San Antonio Spurs of the N.B.A. Testing and vaccination efforts at Hard Rock Stadium in Miami were temporarily suspended on Monday to allow the college football championship game between Alabama and Ohio State to be played there.
The pool of people eligible for the vaccine in New York has recently expanded to include teachers and a range of other essential workers, as well as any resident who is 65 or older. At first, the vaccine was limited to frontline health care workers and nursing home residents.
The CitiField location is part of New York City’s initiative to establish mass inoculation sites in each of the city’s five boroughs. Vaccination centers opened in Brooklyn and the Bronx this week; locations in Manhattan and Staten Island have not yet been announced.
More than 26,000 vaccine doses were administered in the city on Monday, according to Mr. de Blasio, who is trying step up the pace of inoculations. The mayor has said his goal is to have one million doses administered by the end of January.
Mr. Cuomo, a third-term Democrat, said on Tuesday that the state intended to set up a series of rapid testing sites in areas where restrictions have closed indoor dining and arts events, and closed offices. Some of these sites would be located in vacant retail spaces or shuttered businesses, he said, promising hundreds of “pop-up” testing sites.
At the same time, Mr. Cuomo wants to reopen office buildings — a major element of New York City’s economy, both for their tenants and developers — saying he had received assurances from their owners that they could ramp up testing for workers. “Bringing workers back safely will boost ridership on our mass transit, bring customers back to restaurants and stores, and return life to our streets,” he said.
 A coronavirus testing site in Los Angeles on Monday. The United States was one of the poorest-performing countries in a study of responses to the pandemic.Credit…Alex Welsh for The New York Times
A coronavirus testing site in Los Angeles on Monday. The United States was one of the poorest-performing countries in a study of responses to the pandemic.Credit…Alex Welsh for The New York Times
How well a country has responded to Covid-19 is not explained by the country’s economic power or scientific capacity, but by how its people relate to one another and their government, according to preliminary findings of a research study.
“Countries with traditions of acting in concert against social problems, and countries with histories of deference to public authorities, fared better on compliance than countries lacking either or both,” the researchers wrote.
Investigators compared characteristics of 23 countries on six continents, considering outcomes related to disease burden, economic impact and disparities. In the United States, rated as one of the poorest-performing countries, “the virus ‘exploited’ pre-existing weaknesses” in public health, the economy and politics.
Before the pandemic, numerous reports and congressional testimony “recognized vulnerabilities that became apparent during Covid-19,” another study found, including threats of viruses emerging from animals, economic disruption, inadequate stockpiles and vulnerability to global supply shortages. For that study, researchers compiled more than 1,200 pre-pandemic records in an expanding online library that was introduced on Tuesday — Health Security Net — in the hopes that it will “inform future planning and response efforts.”
Another team, studying five countries in Africa, found that national leaders there had quickly recognized the threat from the virus and imposed measures to limit its importation and spread. “That managed to at least curtail the outbreak,” said Wilmot James, a Columbia University research scholar who was one of the study’s principal investigators, “but the impacts on the economies were quite devastating.”
The Africa Centers for Disease Control and Prevention, a four-year-old institution modeled in part on its U.S. counterpart, was unique in providing technical assistance for an entire continent.
The research reports were released Tuesday in conjunction with a two-day symposium, the Futures Forum on Preparedness, supported by Schmidt Futures and the Social Science Research Council.