Here’s what you need to know:
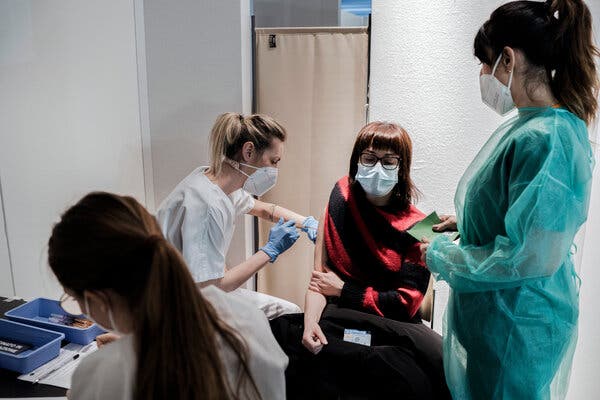
AstraZeneca’s Covid-19 vaccine being administered in Milan on Monday.Credit…Alessandro Grassani for The New York Times
Federal health officials said Tuesday that the encouraging results that AstraZeneca announced about its Covid-19 vaccine may have been based on outdated and incomplete information about the vaccine’s effectiveness, an extraordinary blow to the credibility of an already embattled vaccine.
In a statement released after midnight, the National Institute of Allergy and Infectious Diseases said that an independent panel of medical experts that has been helping to oversee AstraZeneca’s U.S. trial had “expressed concern that AstraZeneca may have included outdated information from that trial, which may have provided an incomplete view of the efficacy data.”
The exact nature of the institute’s concerns — and the effect they might have on how effective the vaccine appears to be — was unclear. But it is highly unusual for such a dust-up about the integrity of a clinical trial, especially one as high-profile as this, to occur in public.
“This is really what you call an unforced error,” Dr. Anthony S. Fauci, the nation’s leading infectious-disease expert, said on “Good Morning America” on Tuesday morning. “Because the fact is: This is very likely a very good vaccine, and this kind of thing does, as you say, do nothing but really cast some doubt about the vaccines and maybe contributes to the hesitancy.”
AstraZeneca defended the data that it released on Monday, which showed the vaccine was 79 percent effective at preventing Covid-19. The company said in a statement on Tuesday that the interim results, which were current as of Feb. 17, appeared to be “consistent” with more recent data collected during the trial. AstraZeneca said it would immediately share its latest efficacy data with the monitoring board. The company said it would reissue fuller results within 48 hours.
The results that AstraZeneca announced on Monday seemed encouraging — especially because they came at a moment when concerns about the vaccine’s safety had led more than a dozen countries, mostly in Europe, to temporarily suspend the shot’s use over concerns about possible rare side effects.
But the statement from the National Institute of Allergy and Infectious Diseases, which is part of the National Institutes of Health, raised the prospect that the company was presenting an overly optimistic interpretation of the data.
In recent days, the independent monitoring board’s analysis was delayed several times because the board had to ask for revised reports from those handling trial data on behalf of the company, according to a person familiar with the matter who was not authorized to discuss it publicly.
Companies sponsoring drug or vaccine trials typically wait for the monitoring board to review analyses and conclude that the study has yielded an answer before they announce trial results.
Company executives do not see the results of the study until the monitoring board reports their study data back to the company. The monitoring board ultimately conveyed the results of the study to AstraZeneca in a meeting over the weekend, leading to the company’s announcement Monday morning.
The monitoring board’s slow progress fueled concerns among federal officials that AstraZeneca may have been sitting on the data or that the monitoring board had concerns about the way the data it was reviewing had been presented.
An AstraZeneca spokeswoman, whom the company declined to name, said on Friday that it was “completely incorrect” that the trial data had formatting problems or had not been submitted to the monitoring board in a clean fashion.
“As is often the case,” the spokeswoman said, monitoring boards “can request new or clarifying analyses of data from the trial. This would enable them to ensure the robustness of their determinations.”
The national institute’s statement, issued shortly after midnight, stunned experts. Dr. Eric Topol, a clinical trials expert at Scripps Research in San Diego, said it was “highly irregular” to see such a public display of friction between a monitoring board and a study sponsor, which are typically in close concordance.
“I’ve never seen anything like this,” he said. “It’s so, so troubling.”
AstraZeneca’s relationship with the U.S. authorities has been fraught since last year, when senior health officials believed the company was not being forthright about the design of its clinical trials, its results and safety issues. That skepticism carried over to last week, when senior officials at a number of federal health agencies grew suspicious about why AstraZeneca had not announced data from its U.S. study.
| United States › |
United StatesOn March 22 |
14-day change |
| New cases |
55,621 |
–8% |
|
| New deaths |
650 |
–35% |
|
| World › |
WorldOn March 22 |
14-day change |
| New cases |
416,353 |
+25% |
|
| New deaths |
7,301 |
+3% |
|
U.S. vaccinations ›

 Munich last week. The number of coronavirus cases in Germany is rising, prompting the government to extend lockdown measures.Credit…Laetitia Vancon for The New York Times
Munich last week. The number of coronavirus cases in Germany is rising, prompting the government to extend lockdown measures.Credit…Laetitia Vancon for The New York Times
Chancellor Angela Merkel of Germany, warning on Tuesday that her country is facing a significantly more deadly wave of the coronavirus, announced a five-day lockdown over Easter and the extension of existing restrictions until mid-April in an effort to break a spike in coronavirus cases.
Starting April 1, and until the following Monday, Germany will effectively shut down for an extended Easter break, with private meetings limited to no more than two groups of up to five adults and almost all stores ordered shuttered (supermarkets can open on the Saturday). Churches are asked to hold services online, and people are being asked to stay home and not travel.
“We are in a very, very serious situation,” Ms. Merkel told a news conference, after hours of deliberations with the leaders of the country’s 16 states over the Easter lockdown and extension of existing restrictions through April 18.
“After we were able to sharply bring down the number of new infections in January, we are now experiencing, through the spread of the more contagious British variant, a more dangerous variation — the numbers are going up and the intensive care beds are filling up,” she said.
Germany is the latest country in Europe to tighten restrictions as more contagious virus variants spread and the continent struggles to vaccinate its citizens. Poland, Italy and parts of France have ordered that residents stay home, and many businesses have shut before the holiday.
A resurgent virus and lagging vaccinations have forced governments to renege on promises that they would slowly reopen businesses and society as spring approached. That has spurred protests across Europe.
Europe’s vaccine campaign slowed after a small number of cases of blood clots and abnormal bleeding were reported in patients who received the AstraZeneca vaccine, dampening confidence in its safety. While the European drug regulator, the European Medicines Agency, cleared the vaccine for use last week and said it was “safe and effective,” the scare further complicated vaccination efforts.
Just three weeks ago, Ms. Merkel and state officials hammered out a road map to reopening that relied on a decline in case rates. But the number of new daily cases in Germany has increased by 69 percent in the past two weeks, to levels last seen in January.
 Regeneron’s monoclonal antibody treatment sharply cuts the risk of hospitalization and death among high-risk Covid-19 patients, a study found.Credit…Regeneron, via Associated Press
Regeneron’s monoclonal antibody treatment sharply cuts the risk of hospitalization and death among high-risk Covid-19 patients, a study found.Credit…Regeneron, via Associated Press
A monoclonal antibody treatment developed by the drug maker Regeneron sharply cut the risk of hospitalization and death when given to high-risk Covid-19 patients in a large clinical trial, the company announced on Tuesday.
The results are the latest in a growing flurry of evidence that the infused drugs, meant to mimic the antibodies that the immune system generates naturally in fighting the coronavirus, can help infected patients avoid the worst outcomes if given early.
Regeneron’s treatment, a cocktail of two antibody drugs, was given last fall to President Donald J. Trump shortly after he got sick with Covid-19 and is now one of three such therapies available in the United States.
The new results come from a Phase 3 trial that enrolled more than 4,500 patients beginning in late September, around the time virus cases began to climb dangerously in the United States. The study found that patients who got the infused treatment within 10 days of developing symptoms or testing positive had a roughly 70 percent reduced risk of being hospitalized or dying compared with patients who were infused with a placebo.
“I think these are exciting data,” said Dr. Rajesh Gandhi, an infectious diseases physician at Massachusetts General Hospital who was not involved in the study.
Even as vaccinations speed up, antibody treatments are expected to be helpful for high-risk people who still get sick for many months at least, and longer still if the virus can’t be wiped out. While there are signs that emerging virus variants may in some cases make antibodies less potent, Regeneron’s cocktail has not shown such vulnerability in laboratory tests.
In the new findings, Regeneron’s treatment worked equally well when given at half the dosing at which it was authorized. Regeneron said that it planned to request that the Food and Drug Administration allow the treatment to be given at that reduced strength.
Such a change would bring several advantages: While the cocktail is safe, getting it at a lower dose reduces the odds of side effects, such as an infusion reaction.
It would also allow Regeneron to increase the supply it can provide the United States. The company said that it had expected to supply the country with about 750,000 doses at the originally authorized higher strength by the end of June. If the lower strength is authorized, the company expects to provide about 1.25 million doses by then.
The antibody treatments from Regeneron and the drug maker Eli Lilly, which makes the other two such drugs authorized in the United States, were expected to be in high demand and to serve as a bridge in fighting the pandemic before vaccinations ramped up. Instead, they ended up sitting on refrigerator shelves in many places even during recent surges.
Many patients and their doctors did not know to ask for them or where to find them. Overwhelmed hospitals lacked the bandwidth to prioritize giving out the treatments. And some doctors were unconvinced by the relatively weak evidence available last fall supporting their use.
That picture is gradually shifting, thanks to improved logistics and more awareness. And more solid evidence, like the new data from Regeneron, also appears to be helping the drugs get used more widely. “As the data get stronger and stronger, I would expect that use will increase,” Dr. Gandhi said.
 People enjoying a Friday evening as businesses and restaurants begin to reopen at Fisherman’s Wharf in San Francisco this month.Credit…Jim Wilson/The New York Times
People enjoying a Friday evening as businesses and restaurants begin to reopen at Fisherman’s Wharf in San Francisco this month.Credit…Jim Wilson/The New York Times
Positive trends in pandemic statistics in the United States are easy to distrust. After all, the country went through two false dawns last year, in the late spring and then again in the late summer, when declines in case reports prefaced even darker days. Each time, the apparent good news prompted relaxations and reopenings that helped bring on the next wave.
So it is no surprise that public health experts are wary about the latest flattening in the curve of the pandemic, from the steep decline in cases seen in late January and February to something like a plateau or slight decline more recently. With more contagious virus variants becoming prevalent, they fear the good news could be ending and a fourth wave might be building.
On Monday, Dr. Rochelle Walensky, the director of the Centers for Disease Control and Prevention, again warned Americans about the spread of the coronavirus, saying that with increased travel, looser pandemic restrictions and worrisome variants bearing down on the United States, another surge could erupt if Americans did not take protection efforts seriously “for just a little bit longer.”
“We are at a critical point in this pandemic, a fork in the road, where we as a country must decide which path we are going to take. We must act now,” said Dr. Walensky, who has been one of many federal officials in recent weeks to warn governors against lifting mask mandates too soon. “And I am worried that if we don’t take the right actions now, we will have another avoidable surge, just as we are seeing in Europe right now and just as we are so aggressively scaling up vaccination.”
That said, there are positive signs:
-
Daily death reports, which stayed stubbornly high long after the post-holidays surge, have finally come down sharply, to levels not seen since mid-November. As of Monday, the nation had averaged 1,051 newly reported virus deaths a day over the past week; the average had hovered around 3,000 for weeks over the winter.
-
Some recent hot spots have made major progress — notably Los Angeles, whose mayor, Eric Garcetti, said on CBS on Sunday that he had “not felt this optimism in 12 months.” The city and surrounding county, where cases in some areas leapt 450 percent over the holidays and hospitals became so swamped that some turned away ambulances, now has a test positivity rate of about 1.9 percent, and in an important shift, new case reports have fallen among people experiencing homelessness.
-
Vaccinations are becoming more accessible by the week, as states receive more doses and open up eligibility, in some cases to include all adult residents. The number of doses administered nationwide each day is rising, and the country surpassed President Biden’s initial goal to have administered 100 million shots on March 19, almost six weeks ahead of schedule.
The question now is which will prevail: the positive effects of trends like these or the negative effects of looser behavior and the evolution of the virus into more dangerous forms?
It’s still “a race between vaccinations and variants,” Dr. Ashish Jha, dean of the Brown University School of Public Health, said on Twitter. Like other experts, he cautioned: “Opening up too fast helps the variants.”
Noah Weiland contributed reporting.
 Samar Khan expected to recover fully from a mild case of Covid-19, but before long her symptoms multiplied, including a “really intense brain fog.”Credit…Taylor Glascock for The New York Times
Samar Khan expected to recover fully from a mild case of Covid-19, but before long her symptoms multiplied, including a “really intense brain fog.”Credit…Taylor Glascock for The New York Times
In the fall, after Samar Khan came down with a mild case of Covid-19, she expected to recover and return to her previous energetic life in Chicago. She was 25 and healthy.
But weeks later, she said, “this weird constellation of symptoms began to set in.”
She had blurred vision encircled with halos. She had ringing in her ears, and everything began to smell like cigarettes or Lysol. One leg started to tingle, and her hands would tremble while she was putting on eyeliner.
She also developed “really intense brain fog,” she said. Trying to concentrate on a call for her job in financial services, she felt as if she had come out of anesthesia.
By the end of the year, Ms. Khan was referred to a special clinic for Covid-related neurological symptoms at Northwestern Memorial Hospital in Chicago, which has been evaluating and counseling hundreds of people with similar problems.
Now, the clinic has published the first study focused on long-term neurological symptoms in people who were never physically sick enough from Covid-19 to need hospitalization, including Ms. Khan.
The study of 100 patients from 21 states, published on Tuesday in The Annals of Clinical and Translational Neurology, found that 85 percent of them experienced four or more neurological issues like brain fog, headaches, tingling, muscle pain and dizziness.
“We are seeing people who are really highly, highly functional individuals, used to multitasking all the time and being on top of their game, but, all of a sudden, it’s really a struggle for them,” said Dr. Igor J. Koralnik, the chief of neuro-infectious diseases and global neurology at Northwestern Medicine, who oversees the clinic and is the senior author of the study.
 City Hall Park and Tweed Courthouse in Downtown Manhattan.Credit…Jose A. Alvarado Jr. for The New York Times
City Hall Park and Tweed Courthouse in Downtown Manhattan.Credit…Jose A. Alvarado Jr. for The New York Times
With virus cases seeming to stabilize in New York City and vaccinations becoming more widespread, city officials intend to send a message that New York is close to returning to normal: On May 3, the city will compel its municipal office employees to begin to report to work in person, according to planning documents shared with The New York Times. Workers will return in phases over several weeks.
Mayor Bill de Blasio’s decision to bring the nation’s largest municipal work force back to the office signals a remarkable turnabout in the fortunes of a city that was the national epicenter of the pandemic, coming to symbolize the perils of living in densely packed global capitals.
The move is meant to broadcast that New York City will soon be open for business, and to encourage private companies to follow suit.
The new policy is expected to affect about 80,000 employees who have been working remotely, including caseworkers, computer specialists and clerical associates. The rest of the city’s roughly 300,000-person work force, many of them uniformed personnel including police officers, firefighters and sanitation workers, have already been reporting to work sites.
“Above all else, this is a major momentum builder,” said Reggie Thomas, a senior vice president with the Real Estate Board of New York.
Yet the move has spurred concern among some workers and union leaders who fear it is premature. New York City still has among the highest coronavirus case rates in the nation. Many workers will have to commute an hour or more on mass transit.
Facial masks will be strongly encouraged but not required: A March 18 presentation from the city’s Department of Citywide Administrative Services said agency leaders should “encourage face coverings to be worn at all times even if six-feet distancing can be maintained.” The provision allows workers to remove face coverings if they are more than six feet apart.
Vaccination will not be mandatory for those returning to the office because of legal concerns, though city officials are strongly encouraging their workers to get vaccinated and are trying to facilitate that process.
 At Heathrow Airport, near London, last month. England’s new rules would exclude those traveling for some work, elite sporting competitions or education.Credit…Henry Nicholls/Reuters
At Heathrow Airport, near London, last month. England’s new rules would exclude those traveling for some work, elite sporting competitions or education.Credit…Henry Nicholls/Reuters
Residents of England who travel abroad without a valid reason will be fined 5,000 pounds, or $6,900, under coronavirus regulations that are scheduled to come into force on Monday if lawmakers approve.
Daily coronavirus deaths in Britain have dropped to their lowest level since fall, thanks in part to a vaccination program that has already reached more than half the adult population, and the country is preparing to slowly reopen its economy after months of national lockdown. A stay-at-home order is to be lifted on Monday, though many shops and other businesses will be closed until mid-April or later.
Travel abroad for leisure is banned until May 17 at the earliest, and the new regulations signal a potentially longer wait for vacationers.
If the new regulations are approved, travelers would have to provide a valid excuse for leaving the country, which would include some essential work, elite sports competitions and education. But opposition lawmakers have criticized an exemption that would allow travel “in connection with the purchase, sale, letting or rental of a residential property,” arguing that it would privilege those wealthy enough to own a second residence. Travel without an essential reason is also banned in Scotland, Wales and Northern Ireland.
The legislation, which is set to be reviewed on April 12 and expire at the end of June, would also renew a ban on indoor gatherings and limit outdoor gatherings to six people. Lawmakers on Thursday will also vote on extending a coronavirus act that gave the government emergency powers during the pandemic, which has caused friction among some members of the governing Conservative Party who have called the laws extreme.
It comes as the country marks the one year since Prime Minister Boris Johnson announced the first national lockdown. Britain has reported at least 4.3 million cases and over 126,000 deaths according to a New York Times database.
 The Regal Cinemas theater in Times Square. The theater chain’s parent company, Cineworld.Credit…Nathan Bajar for The New York Times
The Regal Cinemas theater in Times Square. The theater chain’s parent company, Cineworld.Credit…Nathan Bajar for The New York Times
Cineworld, the parent company of the U.S. movie theater chain Regal Cinemas, announced on Tuesday that it would reopen its cinemas in the United States in April and in Britain in May as those countries ease lockdown restrictions.
“We have long-awaited this moment,” said Mooky Greidinger, the chief executive of Cineworld, which is based in London. “With capacity restrictions expanding to 50 percent or more across most U.S. states, we will be able to operate profitably in our biggest markets.”
Regal Cinemas is the second largest theater chain in the United States, after AMC Theaters. The announcement by Cineworld comes six months after the movie theater chains were forced to shut down across the United States and Britain last October in an effort to curb the spread of the coronavirus. The decision affected a total of 45,000 employees in both countries and forced studios to postpone film releases.
Cineworld also announced a multiyear agreement with Warner Bros. starting in 2022 that will allow the theater chain to show the studios’ films for 45 days in the United States and 31 days in Britain. The deal shortens the typical window that theaters have to show movies before they are released to on-demand streaming services.
The reopening plans in the United States will coincide with the release of two movies from Warner Bros. Pictures, “Godzilla vs. Kong” on April 2 and “Mortal Kombat” on April 16.
“We are very happy for the agreement with Warner Bros.,” Mr. Greidinger said. “This agreement shows the studio’s commitment to the theatrical business.”
Last week, AMC Theaters announced the reopening of nearly all of its U.S. theaters.
The moves come at a time of concern that looser restrictions will lead to rise in coronavirus cases. On Monday, the director of the Centers for Disease Control and Prevention warned that relaxed pandemic restrictions could lead to another spike. “If we don’t take the right actions now,” said Dr. Rochelle Walensky, “we will have another avoidable surge.”
In September, Cineworld reported a pretax loss of $1.6 billion for the first half of 2020. In 2019, 90 percent of the company’s revenue was generated in the United States and Britain.
 A rally of parents and schoolchildren to re-open the public schools in Scotch Plains-Fanwood at the Board of Education office in Scotch Plains.Credit…James Estrin/The New York Times
A rally of parents and schoolchildren to re-open the public schools in Scotch Plains-Fanwood at the Board of Education office in Scotch Plains.Credit…James Estrin/The New York Times
Most school districts in New Jersey have partly reopened, but one in four children still live in a district where public schools are closed. No state in the Northeast had more districts relying on all-virtual teaching in early March than New Jersey, according to Return to Learn, a database created by a conservative think tank, the American Enterprise Institute, and Davidson College. Nationwide, only seven states had a greater proportion of all-remote instruction.
As the distribution of vaccines has accelerated and President Biden has signaled a push for broader reopenings, frustration among parents has grown, particularly in New Jersey’s affluent suburbs, where schools with stellar reputations are a key reason families are willing to pay some of the nation’s highest taxes.
These parents have filed federal lawsuits, held protests, created online petitions and shown up at virtual board of education meetings to demand expanded in-person instruction.
The pressure to open schools more fully comes as the infection rate in New Jersey, which is small and densely populated, remains stubbornly high: With a weekly average of 45 cases for every 100,000 residents, the state leads the nation in new infections per capita, according to a New York Times database.
The drumbeat intensified after the Centers for Disease Control and Prevention announced a major policy shift on Friday, reducing its distancing recommendations to three feet from six feet for all elementary schools and for middle and high schools in areas where the virus infection rate is not high.
Anger at the pace of reopening has led some families who can afford it to enroll their children in private schools, start home-schooling them or move. If enough children leave a district in New Jersey, it could lead to cuts in state aid, scaled-back programming or potentially layoffs.
Several New Jersey cities and counties have held educator-only vaccine distribution events. But the virus’s hold on the state has left teachers and their powerful unions wary of expanded reopening.
 Testing for Covid-19 at a local market in Mumbai, India, on Tuesday. Credit…Divyakant Solanki/EPA, via Shutterstock
Testing for Covid-19 at a local market in Mumbai, India, on Tuesday. Credit…Divyakant Solanki/EPA, via Shutterstock
Mumbai, India’s financial hub, has begun random testing for the coronavirus in malls, railway stations and other crowded places as officials attempt to tamp down on a worrying surge in cases.
Rapid antigen tests will be taken without individuals’ consent, the Municipal Corporation of Greater Mumbai said in a statement on Monday. Anyone who resists will be in violation of India’s colonial-era epidemic act, which gives the government the power to fine or imprison people who violate rules to contain an outbreak.
“We are trying to implement the existing protocol to the strictest possible level: use of face mask, regulating the number of people in one event, use of hand sanitizer, and now tests,” Suresh Kakani, a senior municipal official in Mumbai, told The New York Times.
Active Covid-19 cases in Mumbai have risen by more than 140 percent since March 1. With variants circulating and commercial activity almost back to prepandemic levels, the number of infections has also shot up in the surrounding state of Maharashtra. An entire district was forced back into lockdown last week.
Mr. Kakani said officials are determined to avert another lockdown in Mumbai, the city of 20 million that is home to Bollywood, India’s film industry, as well as the country’s largest stock exchange.
Another lockdown would be economically disastrous for India, which is just starting to recover from a lockdown last year that triggered a humanitarian crisis, as millions of migrant workers fled cities for their home villages, and a recession.