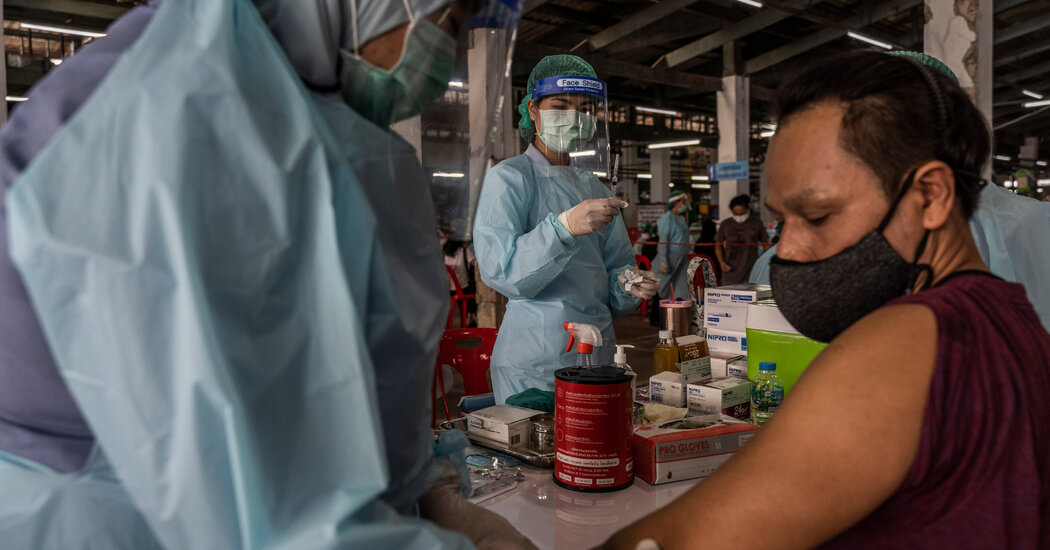
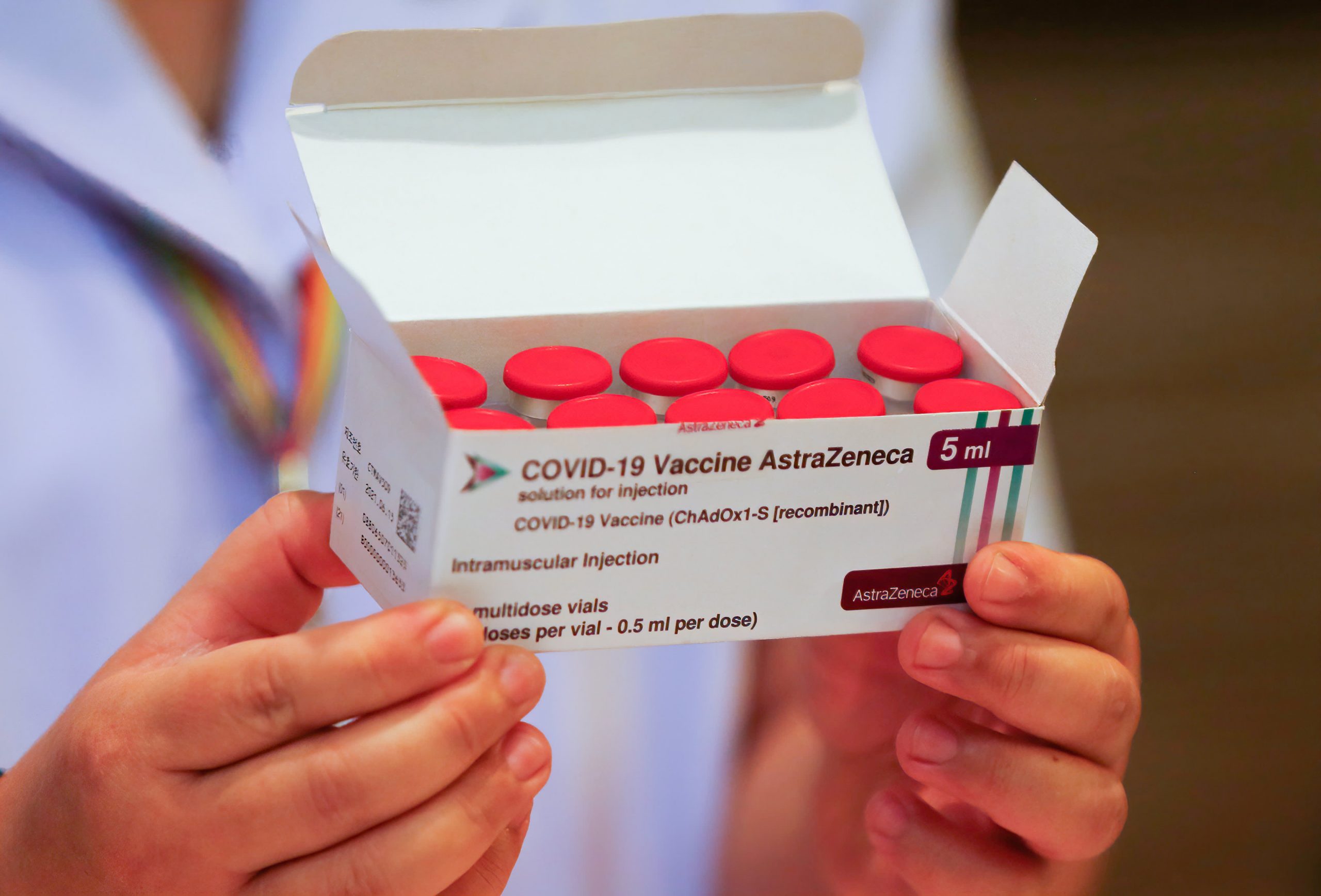
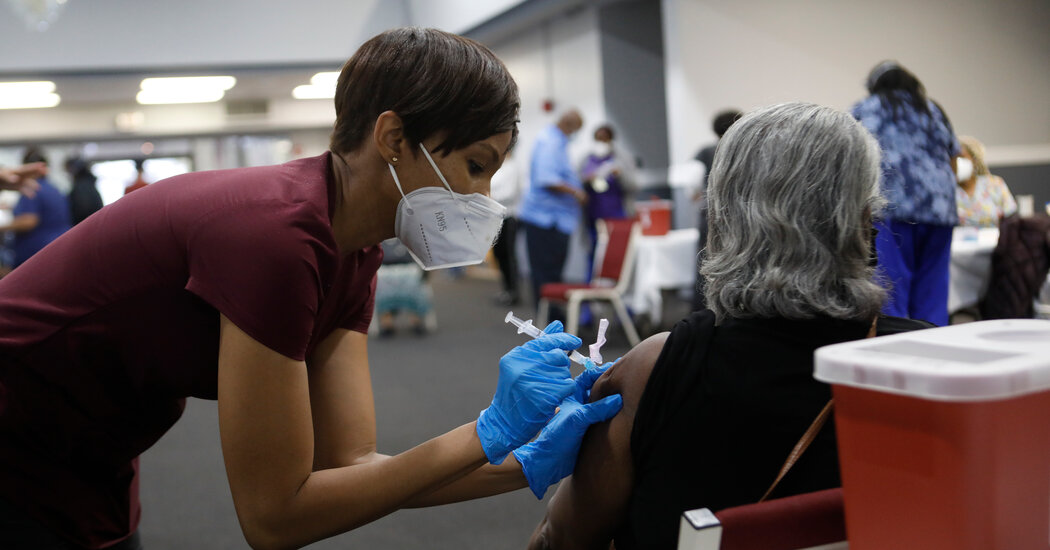
Ein neuer Impfstoff gegen Covid-19, der in Brasilien, Mexiko, Thailand und Vietnam in klinische Studien geht, könnte die Art und Weise verändern, wie die Welt die Pandemie bekämpft. Der Impfstoff mit der Bezeichnung NVD-HXP-S ist der erste in klinischen Studien, der ein neues molekulares Design verwendet, von dem allgemein erwartet wird, dass es wirksamere Antikörper erzeugt als die aktuelle Generation von Impfstoffen. Und der neue Impfstoff könnte viel einfacher herzustellen sein.
Bestehende Impfstoffe von Unternehmen wie Pfizer und Johnson & Johnson müssen in spezialisierten Fabriken unter Verwendung schwer zu beschaffender Inhaltsstoffe hergestellt werden. Im Gegensatz dazu kann der neue Impfstoff in Hühnereiern in Massenproduktion hergestellt werden – dieselben Eier, die jedes Jahr in Fabriken auf der ganzen Welt Milliarden von Influenza-Impfstoffen produzieren.
Wenn sich NVD-HXP-S als sicher und wirksam erweist, könnten Grippeimpfstoffhersteller möglicherweise weit über eine Milliarde Dosen davon pro Jahr produzieren. Länder mit niedrigem und mittlerem Einkommen, die derzeit Schwierigkeiten haben, Impfstoffe aus wohlhabenderen Ländern zu erhalten, können möglicherweise NVD-HXP-S für sich selbst herstellen oder es zu geringen Kosten von Nachbarn erwerben.
“Das ist atemberaubend – es würde das Spiel verändern”, sagte Andrea Taylor, stellvertretende Direktorin des Duke Global Health Innovation Center.
Zunächst müssen klinische Studien jedoch nachweisen, dass NVD-HXP-S tatsächlich bei Menschen wirkt. Die erste Phase der klinischen Studien wird im Juli abgeschlossen sein, und die letzte Phase wird noch einige Monate dauern. Experimente mit geimpften Tieren haben jedoch Hoffnungen auf die Aussichten des Impfstoffs geweckt.
“Es ist ein Heimrennen zum Schutz”, sagte Dr. Bruce Innis vom PATH Center for Vaccine Innovation and Access, das die Entwicklung von NVD-HXP-S koordiniert hat. “Ich denke, es ist ein Weltklasse-Impfstoff.”
2P zur Rettung
Impfstoffe wirken, indem sie das Immunsystem gut genug mit einem Virus bekannt machen, um eine Abwehr dagegen zu veranlassen. Einige Impfstoffe enthalten ganze Viren, die abgetötet wurden. andere enthalten nur ein einziges Protein aus dem Virus. Wieder andere enthalten genetische Anweisungen, mit denen unsere Zellen das virale Protein herstellen können.
Sobald das Immunsystem einem Virus oder einem Teil davon ausgesetzt ist, kann es lernen, Antikörper herzustellen, die es angreifen. Immunzellen können auch lernen, infizierte Zellen zu erkennen und zu zerstören.
Im Falle des Coronavirus ist das beste Ziel für das Immunsystem das Protein, das seine Oberfläche wie eine Krone bedeckt. Das als Spike bekannte Protein bindet sich an die Zellen und lässt das Virus dann mit ihnen fusionieren.
Die einfache Injektion von Coronavirus-Spike-Proteinen in Menschen ist jedoch nicht der beste Weg, um sie zu impfen. Das liegt daran, dass Spike-Proteine manchmal die falsche Form annehmen und das Immunsystem dazu veranlassen, die falschen Antikörper herzustellen.
Diese Erkenntnis entstand lange vor der Covid-19-Pandemie. Im Jahr 2015 trat ein weiteres Coronavirus auf, das eine tödliche Form der Lungenentzündung namens MERS verursachte. Jason McLellan, damals Strukturbiologe an der Geisel School of Medicine in Dartmouth, und seine Kollegen machten sich daran, einen Impfstoff dagegen herzustellen.
Sie wollten das Spike-Protein als Ziel verwenden. Aber sie mussten damit rechnen, dass das Spike-Protein ein Formwandler ist. Während sich das Protein auf die Fusion mit einer Zelle vorbereitet, verzieht es sich von einer tulpenartigen Form zu etwas, das eher einem Speer ähnelt.
Wissenschaftler nennen diese beiden Formen die Präfusions- und Postfusionsformen der Spitze. Antikörper gegen die Präfusionsform wirken stark gegen das Coronavirus, aber Postfusionsantikörper stoppen es nicht.
Dr. McLellan und seine Kollegen verwendeten Standardtechniken, um einen MERS-Impfstoff herzustellen, endeten jedoch mit vielen Postfusionsspitzen, die für ihre Zwecke unbrauchbar waren. Dann entdeckten sie einen Weg, das Protein in einer tulpenartigen Präfusionsform zu halten. Alles, was sie tun mussten, war, zwei von mehr als 1.000 Bausteinen im Protein in eine Verbindung namens Prolin umzuwandeln.
Die resultierende Spitze – 2P genannt – für die beiden darin enthaltenen neuen Prolinmoleküle nahm mit weit größerer Wahrscheinlichkeit die gewünschte Tulpenform an. Die Forscher injizierten die 2P-Spikes in Mäuse und stellten fest, dass die Tiere Infektionen des MERS-Coronavirus leicht abwehren konnten.
Das Team meldete ein Patent für seinen modifizierten Spike an, aber die Welt nahm die Erfindung kaum zur Kenntnis. Obwohl MERS tödlich ist, ist es nicht sehr ansteckend und hat sich als relativ geringe Bedrohung erwiesen. weniger als 1.000 Menschen sind an MERS gestorben, seit es zum ersten Mal beim Menschen aufgetreten ist.
Ende 2019 tauchte jedoch ein neues Coronavirus, SARS-CoV-2, auf und begann, die Welt zu verwüsten. Dr. McLellan und seine Kollegen traten in Aktion und entwarfen einen 2P-Spike, der nur für SARS-CoV-2 gilt. Innerhalb weniger Tage nutzte Moderna diese Informationen, um einen Impfstoff für Covid-19 zu entwickeln. Es enthielt ein genetisches Molekül namens RNA mit den Anweisungen zur Herstellung des 2P-Spikes.
Andere Unternehmen folgten bald diesem Beispiel, nahmen 2P-Spikes für ihre eigenen Impfstoffdesigns an und begannen mit klinischen Studien. Alle drei bisher in den USA zugelassenen Impfstoffe – von Johnson & Johnson, Moderna und Pfizer-BioNTech – verwenden den 2P-Spike.
Andere Impfstoffhersteller verwenden es ebenfalls. Novavax hat in klinischen Studien starke Ergebnisse mit dem 2P-Anstieg erzielt und wird voraussichtlich in den nächsten Wochen bei der Food and Drug Administration eine Genehmigung für den Notfall beantragen. Sanofi testet auch einen 2P-Spike-Impfstoff und geht davon aus, dass die klinischen Studien noch in diesem Jahr abgeschlossen sein werden.
Zwei Prolinen sind gut; Sechs sind besser
Dr. McLellans Fähigkeit, lebensrettende Hinweise in der Struktur von Proteinen zu finden, hat ihm tiefe Bewunderung in der Impfstoffwelt eingebracht. “Dieser Typ ist ein Genie”, sagte Harry Kleanthous, Senior Program Officer bei der Bill & Melinda Gates Foundation. “Er sollte stolz auf diese große Sache sein, die er für die Menschheit getan hat.”
Aktualisiert
5. April 2021, 4:37 Uhr ET
Aber als Dr. McLellan und seine Kollegen den 2P-Spike an Impfstoffhersteller weitergaben, wandte er sich für eine genauere Betrachtung wieder dem Protein zu. Wenn der Austausch von nur zwei Prolinen einen Impfstoff verbessern würde, könnten zusätzliche Optimierungen ihn sicherlich noch weiter verbessern.
“Es war sinnvoll, einen besseren Impfstoff zu versuchen”, sagte Dr. McLellan, der jetzt Associate Professor an der University of Texas in Austin ist.
Im März schloss er sich mit zwei anderen Biologen der Universität von Texas, Ilya Finkelstein und Jennifer Maynard, zusammen. In ihren drei Labors wurden 100 neue Spikes mit jeweils einem veränderten Baustein erstellt. Mit Mitteln der Gates Foundation testeten sie jeden einzelnen und kombinierten dann die vielversprechenden Änderungen bei neuen Spikes. Schließlich schufen sie ein einziges Protein, das ihren Wünschen entsprach.
Der Gewinner enthielt die zwei Prolinen in der 2P-Spitze sowie vier zusätzliche Prolinen, die an anderer Stelle im Protein gefunden wurden. Dr. McLellan nannte den neuen Spike HexaPro zu Ehren seiner insgesamt sechs Prolinen.
Die Struktur von HexaPro war sogar stabiler als die von 2P, stellte das Team fest. Es war auch widerstandsfähig, besser in der Lage, Hitze und schädlichen Chemikalien zu widerstehen. Dr. McLellan hoffte, dass sein robustes Design es in einem Impfstoff wirksam machen würde.
Dr. McLellan hoffte auch, dass Impfstoffe auf HexaPro-Basis mehr von der Welt erreichen würden – insbesondere Länder mit niedrigem und mittlerem Einkommen, die bisher nur einen Bruchteil der Gesamtverteilung der Impfstoffe der ersten Welle erhalten haben.
“Der Anteil der Impfstoffe, die sie bisher erhalten haben, ist schrecklich”, sagte Dr. McLellan.
Zu diesem Zweck hat die University of Texas eine Lizenzvereinbarung für HexaPro getroffen, die es Unternehmen und Labors in 80 Ländern mit niedrigem und mittlerem Einkommen ermöglicht, das Protein in ihren Impfstoffen zu verwenden, ohne Lizenzgebühren zu zahlen.
In der Zwischenzeit suchten Dr. Innis und seine Kollegen bei PATH nach einer Möglichkeit, die Produktion von Covid-19-Impfstoffen zu steigern. Sie wollten einen Impfstoff, den weniger wohlhabende Nationen selbst herstellen können.
Mit ein wenig Hilfe von Eiern
Die erste Welle zugelassener Covid-19-Impfstoffe erfordert spezielle, kostspielige Inhaltsstoffe. Zum Beispiel benötigt der RNA-basierte Impfstoff von Moderna genetische Bausteine, sogenannte Nukleotide, sowie eine maßgeschneiderte Fettsäure, um eine Blase um sie herum aufzubauen. Diese Inhaltsstoffe müssen in eigens dafür errichteten Fabriken zu Impfstoffen verarbeitet werden.
Die Art und Weise, wie Influenza-Impfstoffe hergestellt werden, ist dagegen eine Studie. In vielen Ländern gibt es riesige Fabriken für billige Grippeschutzimpfungen, in die Hühnereier mit Influenzaviren injiziert werden. Die Eier produzieren eine Fülle neuer Kopien der Viren. Fabrikarbeiter extrahieren dann die Viren, schwächen oder töten sie und setzen sie dann in Impfstoffe ein.
Das PATH-Team fragte sich, ob Wissenschaftler einen Covid-19-Impfstoff herstellen könnten, der billig in Hühnereiern angebaut werden könnte. Auf diese Weise könnten dieselben Fabriken, die Grippeschutzimpfungen durchführen, auch Covid-19-Impfungen durchführen.
In New York wusste ein Team von Wissenschaftlern der Icahn School of Medicine am Mount Sinai, wie man einen solchen Impfstoff mit einem Vogelvirus namens Newcastle Disease Virus herstellt, das beim Menschen harmlos ist.
Seit Jahren experimentieren Wissenschaftler mit dem Newcastle-Virus, um Impfstoffe für eine Reihe von Krankheiten zu entwickeln. Um beispielsweise einen Ebola-Impfstoff zu entwickeln, fügten die Forscher dem eigenen Satz von Genen des Newcastle-Disease-Virus ein Ebola-Gen hinzu.
Die Wissenschaftler setzten dann das manipulierte Virus in Hühnereier ein. Da es sich um ein Vogelvirus handelt, vermehrte es sich schnell in den Eiern. Die Forscher hatten Viren der Newcastle-Krankheit, die mit Ebola-Proteinen beschichtet waren.
Am Berg Sinai machten sich die Forscher daran, dasselbe zu tun, indem sie Coronavirus-Spike-Proteine anstelle von Ebola-Proteinen verwendeten. Als sie von Dr. McLellans neuer HexaPro-Version erfuhren, fügten sie dies den Newcastle-Krankheitsviren hinzu. Die Viren waren voller Spike-Proteine, von denen viele die gewünschte Präfusionsform hatten. In Anspielung auf das Newcastle-Virus und den HexaPro-Spike nannten sie es NDV-HXP-S.
PATH veranlasste die Herstellung von Tausenden von Dosen NDV-HXP-S in einer vietnamesischen Fabrik, in der normalerweise Influenza-Impfstoffe in Hühnereiern hergestellt werden. Im Oktober schickte die Fabrik die Impfstoffe nach New York, um sie zu testen. Die Forscher des Mount Sinai fanden heraus, dass NDV-HXP-S Mäusen und Hamstern einen starken Schutz verleiht.
“Ich kann ehrlich sagen, dass ich jeden Hamster, jede Maus auf der Welt vor SARS-CoV-2 schützen kann”, sagte Dr. Peter Palese, der Leiter der Forschung. “Aber die Jury ist sich immer noch nicht sicher, was sie beim Menschen tut.”
Die Wirksamkeit des Impfstoffs brachte einen zusätzlichen Vorteil: Die Forscher benötigten weniger Viren für eine wirksame Dosis. Ein einzelnes Ei kann fünf bis 10 Dosen NDV-HXP-S ergeben, verglichen mit einer oder zwei Dosen Influenza-Impfstoffen.
“Wir freuen uns sehr darüber, weil wir glauben, dass dies ein Weg ist, einen billigen Impfstoff herzustellen”, sagte Dr. Palese.
PATH verband dann das Mount Sinai-Team mit Influenza-Impfstoffherstellern. Am 15. März gab das vietnamesische Institut für Impfstoffe und medizinische Biologika den Beginn einer klinischen Studie mit NDV-HXP-S bekannt. Eine Woche später folgte Thailands Government Pharmaceutical Organization. Am 26. März kündigte das brasilianische Butantan-Institut die Genehmigung an, eigene klinische Studien mit NDV-HXP-S zu beginnen.
Inzwischen hat das Mount Sinai-Team den Impfstoff auch als intranasales Spray an den mexikanischen Impfstoffhersteller Avi-Mex lizenziert. Das Unternehmen wird klinische Studien starten, um festzustellen, ob der Impfstoff in dieser Form noch wirksamer ist.
Für die beteiligten Nationen war die Aussicht, die Impfstoffe vollständig selbst herzustellen, attraktiv. “Diese Impfstoffproduktion wird von Thailändern für Thailänder hergestellt”, sagte Thailands Gesundheitsminister Anutin Charnvirakul bei der Ankündigung in Bangkok.
In Brasilien hat das Butantan-Institut seine Version von NDV-HXP-S als „brasilianischen Impfstoff“ bezeichnet, der „vollständig in Brasilien hergestellt wird, ohne von Importen abhängig zu sein“.
Frau Taylor vom Duke Global Health Innovation Center war mitfühlend. “Ich konnte verstehen, warum das wirklich so eine attraktive Aussicht wäre”, sagte sie. “Sie waren den globalen Lieferketten ausgeliefert.”
Madhavi Sunder, ein Experte für geistiges Eigentum an der Georgetown Law School, warnte, dass NDV-HXP-S Ländern wie Brasilien nicht sofort helfen würde, da sie sich mit der aktuellen Welle von Covid-19-Infektionen auseinandersetzen. “Wir sprechen nicht über 16 Milliarden Dosen im Jahr 2020”, sagte sie.
Stattdessen wird die Strategie für die langfristige Impfstoffproduktion wichtig sein – nicht nur für Covid-19, sondern auch für andere Pandemien, die in Zukunft auftreten könnten. “Es klingt super vielversprechend”, sagte sie.
In der Zwischenzeit ist Dr. McLellan zum molekularen Zeichenbrett zurückgekehrt, um zu versuchen, eine dritte Version ihres Spikes herzustellen, die noch besser als HexaPro ist.
“Es gibt wirklich kein Ende für diesen Prozess”, sagte er. „Die Anzahl der Permutationen ist nahezu unendlich. Irgendwann müsste man sagen: ‘Dies ist die nächste Generation.’ “