Developing countries looking for coronavirus vaccines now have another reliable option – and China’s reputation as an emerging scientific superpower has just gotten a big boost.
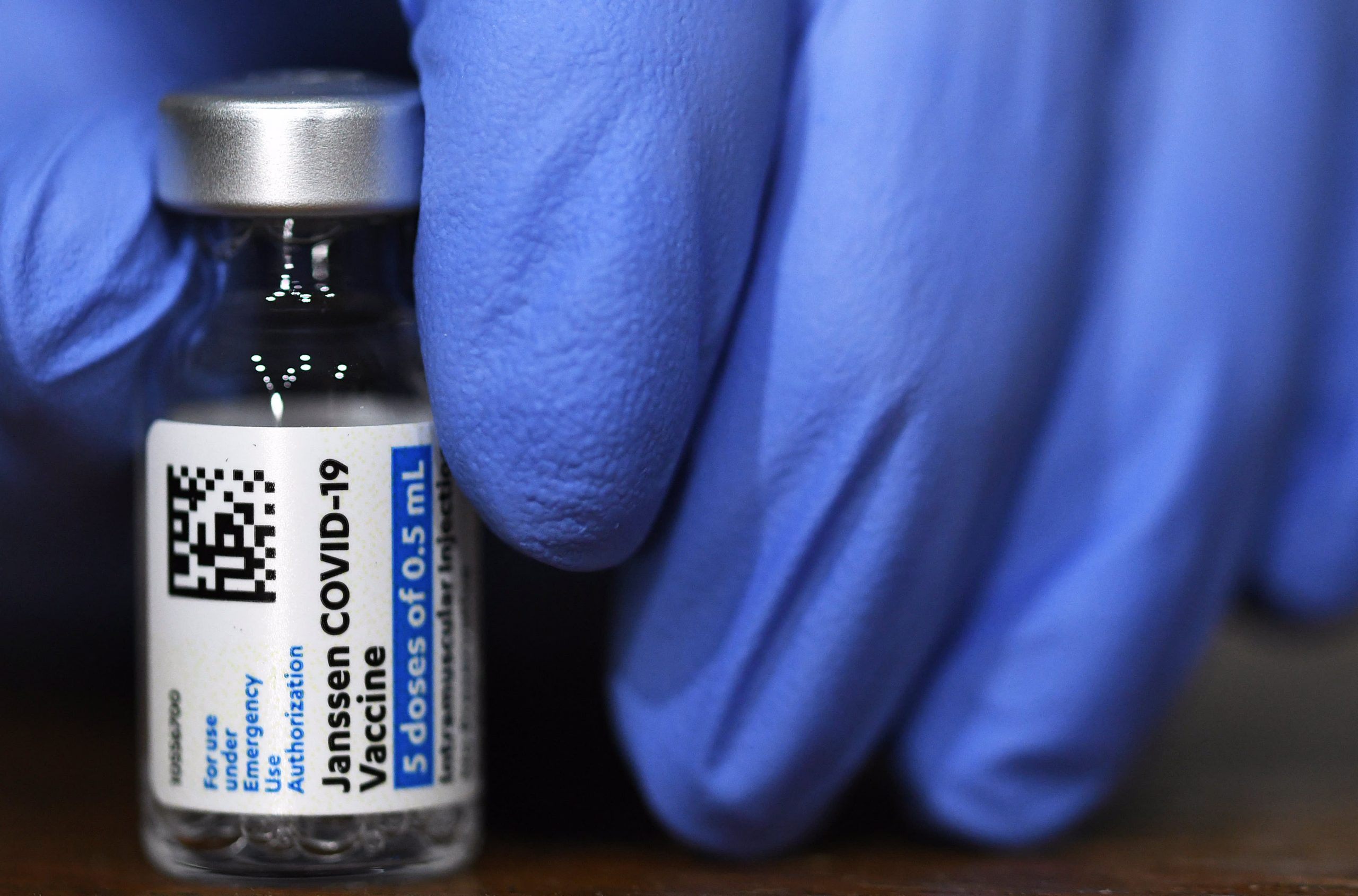
The World Health Organization on Friday declared a vaccine from the Chinese company Sinopharm as a safe and reliable way to fight the virus. The statement marks a significant step in dispelling doubts about the vaccine after the Chinese government and company released little data on late-stage clinical trials.
WHO emergency approval enables Sinopharm vaccine to be included in Covax, a global initiative to provide free vaccines to poor countries. The possible inclusion in Covax raises the hope that more people – especially in developing countries – will have access to recordings at a crucial moment.
Rich countries hoard vaccine doses. India, a major vaccine maker, has stopped exporting to deal with the deepening coronavirus crisis. Safety concerns led health authorities in some countries to temporarily stop using AstraZeneca and Johnson & Johnson vaccines.
“The addition of this vaccine has the potential to quickly accelerate access to Covid-19 vaccines for countries that want to protect health workers and vulnerable populations,” said Dr. Mariângela Simão, WHO Deputy Director General for Access to Health Products, in a statement.
Reliable access to vaccines could improve even further next week if WHO considers another Chinese shot from a company called Sinovac. But the fanfare can be short-lived. While China has claimed it could produce up to five billion cans by the end of this year, Chinese officials say they are struggling to make enough cans for their own people and are warning a pandemic-weary world to keep expectations in check .
“This should be the golden time for China to practice vaccine diplomacy. The problem is also that China itself is facing a shortage, ”said Yanzhong Huang, Senior Fellow on Global Health at the Council on Foreign Relations. “In terms of global access to vaccines, I don’t expect the situation to improve significantly in the next two to three months.”
China’s vaccination campaign got off to a slow start, partly because the government gave export priority and residents did not feel rushed to get vaccinated. The country is now accelerating its national vaccination campaign and aims to vaccinate 40 percent of its 1.4 billion people by the end of June.
Sinopharm and Sinovac are producing about 12 million doses a day, slightly more than the 10 million doses China plans to give daily to meet the domestic target. According to a calculation on data from Bridge Consulting, a Beijing-based consultancy focused on China’s impact on global health, companies would have to produce around 500 million additional doses to meet other countries’ demands.
The vaccine shortage in China underscores the complexity of launching a mass vaccination campaign for the world’s most populous nation and attempting an ambitious export program. Companies involved in the vaccine supply chain, such as syringe manufacturers, work overtime.
Updated
May 7, 2021, 2:53 p.m. ET
“This vaccine is lacking all over the world,” said Pearson Liu, a Sinovac spokesman. “The demand is just too great.”
To make up for the deficit, Chinese officials said those who get vaccinated in China could delay the second shot for up to eight weeks or combine the same type of vaccine from different companies. You said the shortage should subside by June.
Andrea Taylor, who analyzes global data on vaccines at the Duke Global Health Institute, described the possible inclusion of two Chinese vaccines in the Covax program as a “game changer”.
“The current situation is so desperate for low- and lower-middle-income countries that it is worth mobilizing all the doses we can get out of it,” said Ms. Taylor. “Possibly having two options from China could really change the landscape of the possible in the next few months.”
China’s vaccines have been launched in more than 80 countries, but have met with considerable skepticism, partly because the companies have not released data on phase 3 clinical trials to allow scientists to independently evaluate vaccine efficacy rates. A WHO advisory group released the data this week.
According to the WHO advisory group, the Sinopharm vaccine developed with the Beijing Institute of Biological Products has an effectiveness rate of 78.1 percent. The Sinovac vaccine has different efficacy rates between 50 and 84 percent depending on the country in which phase 3 studies were conducted. Both vaccines are made using a proven technology that uses chemicals to weaken or kill a virus.
The advisory group’s data showed that the Sinopharm vaccine had a “high level of confidence” in preventing Covid-19 in adults, but a “low confidence” for people over 60. The group’s results were for the Sinovac vaccine similar .
The WHO said that Sinopharm could not estimate the effectiveness of the vaccine for this group because Sinopharm had only included a few adults over 60 years of age in its studies. However, WHO said it would not restrict use of the vaccine in this age group, as preliminary data suggests that “the vaccine is likely to have protective effects in the elderly”.
There is limited data on how well the vaccine works against the many coronavirus variants that are found around the world. Chinese vaccines are overall less effective than those manufactured by Pfizer-BioNTech and Moderna.
But for China’s leaders, WHO’s approval can still be seen as a badge of honor. Xi Jinping, China’s leader, is committed to making a Covid-19 vaccine a “global public good.”
After India announced export restrictions on vaccines last month, Indonesia and the Philippines said they would turn to China for help. Last week, China’s foreign minister offered to give South Asian countries access to vaccines.
Indonesia said it would receive additional doses of Sinovac after President Joko Widodo held talks with Mr. Xi. In a speech that same week, President Rodrigo Duterte of the Philippines said he owed China “gratitude” for its vaccines.
It remains to be seen whether WHO’s approval will change Beijing’s approach to vaccine distribution. China has only given Covax 10 million doses, despite having independently donated 16.5 million doses and sold 691 million doses to 84 countries, according to Bridge Consulting. Many of the donations went to developing countries in Africa and Asia.
“They don’t like to have their generosity in their products under one UN brand,” said J. Stephen Morrison, director of the global center for health policy at the Center for Strategic and International Studies. “You are in a historic phase,” he said. “They want recipients to know this is China delivering.”
Jason Gutierrez contributed to the coverage. Elsie Chen contributed to reporting and research.