A vial of vaccine. Five elderly homebound patients. Six hours to reach her before the vaccine goes bad.
Doctors at Northwell Health, the largest health care provider in New York State, set out last week to solve one of the toughest medical and logistical challenges facing the campaign to vaccinate Americans against the coronavirus: How to vaccinate the millions of seniors who live there live at home and are too frail or disabled to go to a clinic or line up at a vaccination site.
Members of the network’s home visit program had prepared for their first run. The delivery of Johnson & Johnson’s new coronavirus vaccine made the operation easier as a visit would do the trick.
A medical team designed a route that would include a cluster of homes not far from each other, starting with elderly patients in underserved communities badly affected by the virus. Doctors contacted patients well in advance of the visits, knowing that they would need enough time to consult with their families about the vaccination. Few refused; Most of them loved it.
Before the doctors took to the streets, they checked patients on the phone to make sure they were relatively healthy. Unexpected problems had to be avoided. Doctors ran against the clock: once they broke the vial seal and withdrew the first dose, they only had six hours to use the remaining vaccine or they had to throw it away.
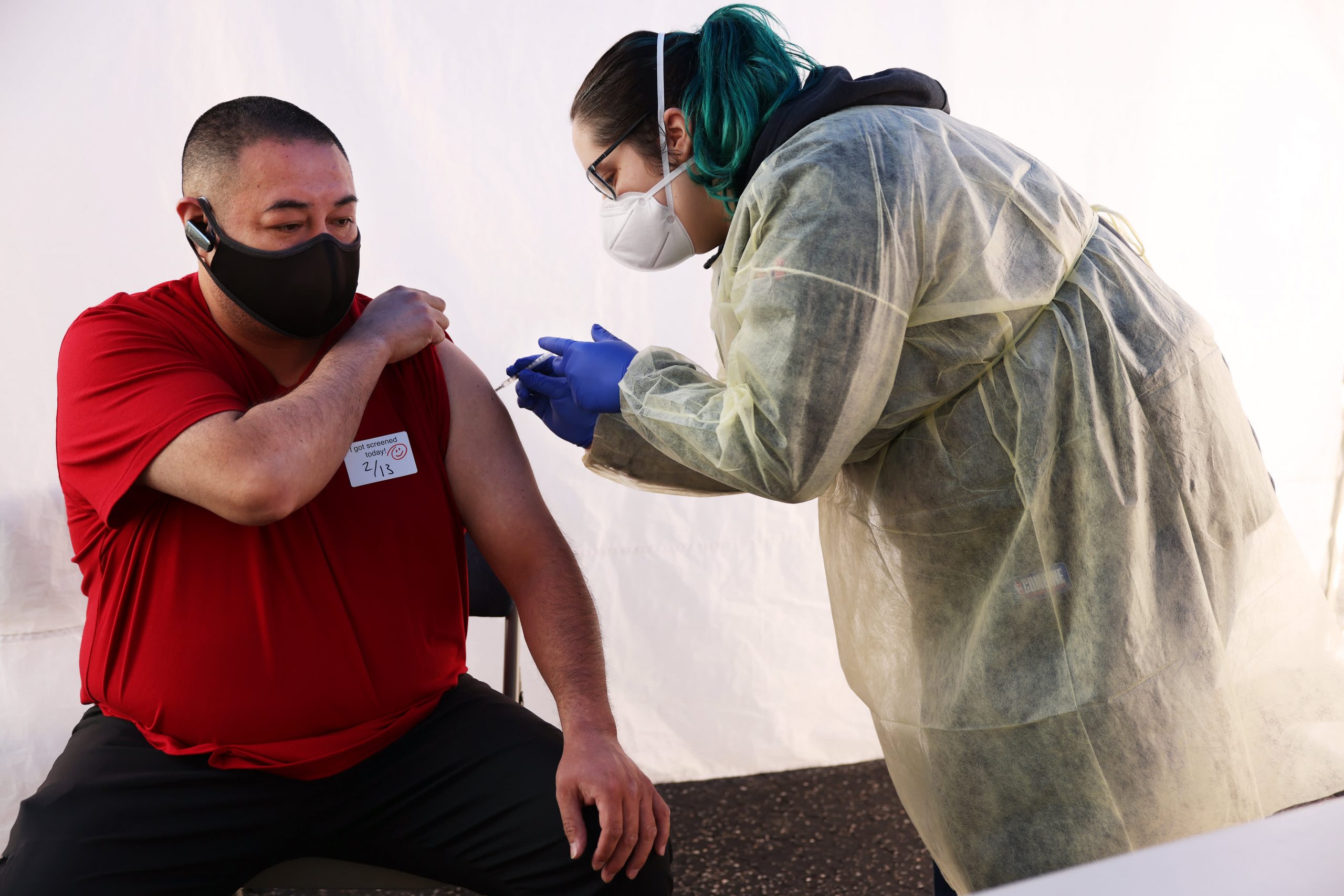
“We’ll be sailing a tight ship, I think, but very compassionately,” said Dr. Karen Abrashkin, the program’s medical director, when a bulky high-tech cooler – actually a car refrigerator – was loaded in the back seat of her car last Wednesday and plugged into a cigarette lighter.
Inside was an ampoule the size of a thimble containing five doses of vaccine. “It’s a historic moment,” she said.
Her first stop was a Twofer, the home of a married couple in Hempstead, New York, Hector Hernandez, 81, a retired window cleaner who used to scrub high-rise buildings in Manhattan, and his wife Irma, 80, a retired seamstress, decided to get vaccinated after reviewing a potpourri of conflicting advice from friends and family.
“At first I was skeptical – is it safe?” Mr. Hernandez said. Two friends had warned him to be careful because the vaccine was new. But Ms. Hernandez’s cardiologist assured the couple it was safe, and another friend seemed confident that it was better to get the vaccine than not to get it.
The couple’s granddaughters, including one who was with Covid-19 for two weeks, advised to wait and see if the vaccine had any long-term side effects. In the end, said Mr Hernandez, her daughter persuaded her to get vaccinated.
“She called and said, ‘You have to make it because if you ever get Covid it can be really bad – you can’t breathe,” said Hernandez.
When Dr. Abrashkin pierced the vial’s seal with a syringe, Lorraine Richardson, a social worker who accompanied her, noted the time: 10:11. The two monitored the Hernandezes for side effects for 15 minutes and then set off. They had until 4:11 p.m. to reach three more patients.
At least two million Americans like the Hernandezes are tied to their homeland, a population that is virtually invisible. Most have multiple chronic conditions but cannot access basic home care. They often end up in hospitals and are prone to the coronavirus because of their ailments.
Updated
March 22, 2021, 8:27 p.m. ET
As public health officials drew up vaccine distribution plans, priority was given to the roughly five million residents and employees of communities such as nursing homes, where the coronavirus spread like wildfire in the early days of the pandemic. The virus killed at least 172,000 residents and employees, accounting for about a third of all deaths from Covid-19 in the United States.
However, the vast majority of Americans over 65 do not live in nursing homes or assisted living facilities, but rather in the community, where they are more difficult to reach. There is no central register for older people in their home country. Geographically dispersed and isolated, they are often difficult to find.
“This could be the next big hurdle for the elderly population,” said Tricia Neuman, senior vice president at the Kaiser Family Foundation. “So much of the vaccination has been a patchwork quilt at the state or local level, but this presents a whole different set of challenges.”
Vaccination rates among seniors have risen rapidly, with at least 60 percent vaccinated so far. Dr. However, Neuman noted that there is no system to get home: “Some people just can’t get to a vaccination site, so the challenge is getting the vaccine to where they live.”
In the absence of a centrally coordinated campaign aimed at home, local initiatives have sprung up across the country. Fire paramedics deliver vaccines to seniors in Miami Beach, Florida and Chicago. A visiting nurse service vaccinates older adults resident in East St. Louis, Illinois as part of the Meals on Wheels program.
Several health systems, like Geisinger Health in Pennsylvania and the Boston Medical Center, have identified hundreds of Americans and sent them vaccines. In Minnesota, nonprofits have set up pop-up vaccination clinics in senior housing and adult daycare.
On Monday, New York City announced that it would expand door-to-door vaccination efforts for seniors in the home, reaching at least 23,000 residents. According to Dr. Linda DeCherrie, the Mount’s clinical director, has the visiting physicians program at Mount Sinai, New York, which serves 1,200 residents, vaccinated 185 patients, and received the green light to vaccinate the senior caregivers of the Sinai at home program as well.
Northwell’s home visit program, which serves patients in Queens, Manhattan, and Long Island, calls for vaccinating 100 patients a week for the next 10 weeks. That schedule could be sped up if nurses were allowed to wear rescue medication in the event patients develop side effects such as anaphylactic shock.
While Dr. Abrashkin administered vaccines in Long Island last week, Dr. Konstantinos Deligiannidis, a colleague, found five elderly women in the Brentwood, NY area within four hours.
“You were so relieved,” he said. “They were all concerned – how could they get the vaccine since they couldn’t get out of the house?”
Dr. Abrashkin and Ms. Richardson visited and vaccinated two more elderly women on Wednesday before making their final stop in the sunny, plant-filled kitchen of Juanita Midgette, 73, a retired computer science and economics teacher with arthritis whom Eddie Murphy counts among her previous students. (Spoiler alert: he was a respectful student, she said, and she recommended his new movie, Coming 2 America.)
It was 12:31 when they knocked on the door. Ms. Midgette had heard mixed reviews of the coronavirus vaccine and had argued with her sister about it. But she hadn’t been able to travel to her North Carolina home and visit relatives since the pandemic, and she hoped the vaccine would give her the freedom to do so.
She believed in God and in science. Ms. Midgette said her research on the vaccine led her to conclude that “positivity far outweighs negativity”.
“My research shows me that with the data they have gathered so far, they are doing the best they can to save lives,” said Ms. Midgette.
“It reminds me of when we had the first computers and they were so big, but we started teaching with them,” she said. “Now they fit in the palm of your hand. If they had waited to get something smaller, the world would look different than it is today. “
After getting the shot, she asked Dr. Abrashkin: “Is it all over?”
“It’s hard to be isolated,” said Ms. Midgette. “I’m looking forward to somehow talking again.”