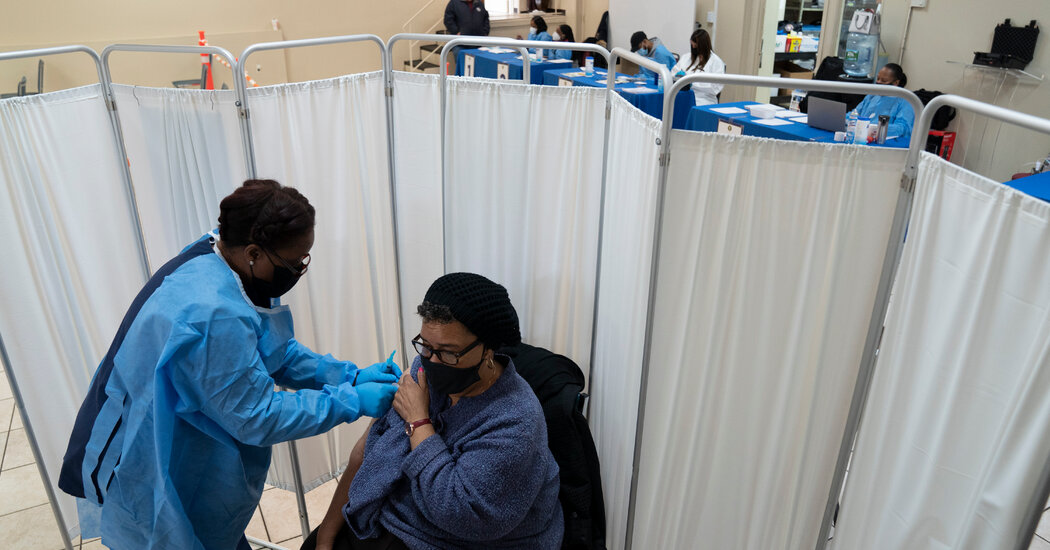
People queue at the bulk vaccination booth at the San Francisco Moscone Convention Center, which opened today on February 5, 2021 in San Francisco, California for healthcare workers and people over 65.
Amy Osborne | AFP | Getty Images
The San Francisco Department of Public Health and Zuckerberg San Francisco General Hospital announced Tuesday that they would allow patients who received the single-dose Covid-19 vaccine from Johnson & Johnson to have a second vaccination from Pfizer-BioNTech or Moderna.
J&J recipients can make special requests to get a “supplementary dose” of an mRNA vaccine, city health officials said in a statement to CNBC, declining to call the second shot a “booster.” J & J’s vaccine only requires one dose and recipients are considered fully vaccinated two weeks after receiving the vaccination.
In a call to reporters later Tuesday, San Francisco health officials said they would allow patients to take the extra syringes due to the high number of requests they received from local residents. They claimed that J & J’s vaccine was highly effective against the virus and its variants.
“We have received requests based on patients speaking to their doctors, so we are allowing the placement,” said Naveena Bobba, assistant director of health for the San Francisco Department of Public Health.
Health officials said they do not currently recommend a booster vaccination, which is in line with guidelines from the Centers for Disease Control and Prevention.
“This step does not represent a policy change for the EVS,” says a statement from the health department. “We are still following the guidelines of the Center for Disease Control and Prevention and currently do not recommend a booster vaccination. We will continue to review all new data and adjust our guidelines if necessary. “
The CDC is currently not recommending that Americans mix Covid vaccinations in most cases, and federal health officials say booster doses of the vaccines are not currently required.
The announcement by the San Francisco health authorities comes as some Americans say they are looking for ways to get extra doses of the Covid vaccines – some even go so far as to get extra vaccinations from various companies – due to concerns about the high contagious delta variant.
Dr. Angela Rasmussen, a virologist at Georgetown University, told CNBC last month that she received a booster of Covid-19 vaccine from Pfizer and BioNTech in late June, two months after receiving the single dose from J&J. She was concerned about her level of protection against Delta after studies showed that a single dose of a Covid vaccine was not enough.
Since Rasmussen received her booster, a new study has found that the J&J vaccine against the Delta and Lambda variants is much less effective than against the original virus. The researchers who led the study, which has not yet been peer-reviewed, are now hoping that J&J recipients will eventually receive a booster of the Pfizer or Moderna vaccines.
Of course, the new research contradicts a study by the company that found the vaccine to be effective against Delta even eight months after vaccination, especially against serious illness and hospitalization. It is likely that the mixing and matching debate in the US will rekindle as the highly contagious Delta variant continues to spread in the US
J&J did not immediately respond to a request for comment on the announcement by the San Francisco Department of Public Health.