Here’s what you need to know:
Empty vials of the Johnson & Johnson vaccine at a hospital in South Africa.Credit…Phill Magakoe/Agence France-Presse — Getty Images
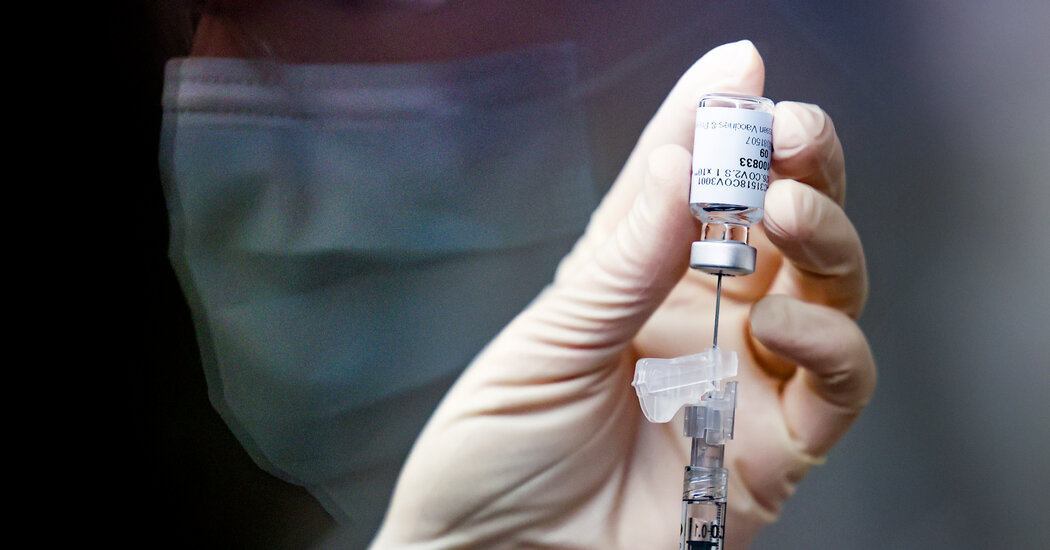
Johnson & Johnson’s Covid-19 vaccine was endorsed on Friday by a panel of experts advising the Food and Drug Administration, clearing the last hurdle before a formal authorization expected on Saturday, according to two people familiar with the agency’s plans. The nation’s first shipments will go out in the days after that.
It will be the third shot made available to the United States in the year since the first surge of coronavirus cases began washing over the country, and it will be the first vaccine to require just one dose instead of two.
Johnson & Johnson’s formulation worked well in clinical trials, particularly against severe disease and hospitalizations, even though it did not match the sky-high efficacy rates of the first two vaccines made by Pfizer-BioNTech and Moderna.
The panel, made up of independent infectious disease experts, statisticians and epidemiologists, voted unanimously in favor of authorizing the vaccine.
“We’re dealing with a pandemic right now,” said Dr. Jay Portnoy, an allergist at Children’s Mercy Hospital in Kansas City, Mo., and a member of the board. “It’s great that we have this vaccine.”
During Johnson & Johnson’s presentation to the panel, Dr. Gregory Poland, a virologist at the Mayo Clinic and a paid external consultant for the company, noted the vaccine’s efficacy, ease of use and low rate of side effects. It “nearly checks all the boxes,” he said. “To me, it is clear that the known benefits vastly outweigh the known risks.”
The vaccine had an overall efficacy rate of 72 percent in the United States and 64 percent in South Africa, where a concerning variant emerged in the fall. The shot showed 86 percent efficacy against severe forms of Covid-19 in the United States, and 82 percent against severe disease in South Africa.
Those are strong numbers, but lower than the roughly 95 percent efficacy rates of Pfizer-BioNTech and Moderna’s vaccines against mild, moderate and severe cases of Covid.
Johnson & Johnson’s vaccine is a single dose and uses a different kind of technology than the authorized vaccines. And the scale and size of the Johnson & Johnson trial was vast, spanning eight countries, three continents and nearly 45,000 participants.
Although the vaccine works with one shot, studies are underway to determine if a second dose would increase its protective effects.
Dr. Paul Offit, a pediatrician at the Children’s Hospital of Philadelphia and one of the panelists, pointed out on Friday that in early clinical trials that took place over the summer, Johnson & Johnson found that a second dose led to levels of coronavirus antibodies that were almost three times higher than those produced by one dose alone.
The results of Johnson & Johnson’s two-dose, late-stage clinical trial are not expected until July at the earliest. If those results turn out to be better than a single dose, Dr. Offit asked, “Does this then become a two-dose vaccine?”
Dr. Johan Van Hoof, the global head of vaccine research and development at Janssen Pharmaceuticals, the drug development arm of Johnson & Johnson, said that the company decided to pursue the one-shot strategy after its studies on monkeys last spring showed that a single dose was enough to provide strong protection against the disease.
“It’s clear that in a situation of an outbreak, in a raging epidemic, the big challenge is to get the epidemic under control,” he said. “The regimen is extremely well positioned to be used in outbreak situations.”
But Dr. Van Hoof also noted that it will be important to track volunteers who received a single dose to see if their immunity changes in the months to come. It might be necessary to deliver a booster shot for long-term protection. “The big question mark still is, how long does protection last?” he said.
After the vote, the F.D.A. told Johnson & Johnson that it “will rapidly work toward finalization and issuance of an emergency use authorization,” according to a statement. The F.D.A. also said that it had notified other government agencies “so they can execute their plans for timely vaccine distribution.”
| United States › |
United StatesOn Feb. 26 |
14-day change |
| New cases |
78,262 |
–29% |
|
| New deaths |
2,266 |
–20% |
|
| World › |
WorldOn Feb. 26 |
14-day change |
| New cases |
410,857 |
–5% |
|
| New deaths |
9,468 |
–23% |
|
U.S. vaccinations ›

Video
transcript
Back
transcript
Decline in Coronavirus Cases ‘May Be Stalling,’ C.D.C. Director Warns
Dr. Rochelle Walensky, director of the Centers for Disease Control and Prevention, said on Friday that a recent decline in coronavirus cases across the U.S. “may be stalling” and urged governors not to relax restrictions.
Over the last few weeks, cases in hospital admissions in the United States have been coming down since early January and deaths have been declining in the past week. But the latest data suggest that these declines may be stalling, potentially leveling off at still a very high number. We at C.D.C. consider this a very concerning shift in the trajectory. We are watching these concerning data very closely to see where they will go over the next few days. But it’s important to remember where we are in the pandemic. Things are tenuous. Now is not the time to relax restrictions. Although we’ve been experiencing large declines in cases and admissions over the past six weeks, these declines follow the highest peak we have experienced in the pandemic. We may be done with the virus, but clearly, the virus is not done with us. We cannot get comfortable or give in to a false sense of security that the worst of the pandemic is behind us. Not now, not when mass vaccination is so very close.
 Dr. Rochelle Walensky, director of the Centers for Disease Control and Prevention, said on Friday that a recent decline in coronavirus cases across the U.S. “may be stalling” and urged governors not to relax restrictions.CreditCredit…Josh Bell/The Sun News, via Associated Press
Dr. Rochelle Walensky, director of the Centers for Disease Control and Prevention, said on Friday that a recent decline in coronavirus cases across the U.S. “may be stalling” and urged governors not to relax restrictions.CreditCredit…Josh Bell/The Sun News, via Associated Press
The federal government warned impatient governors against relaxing pandemic control measures on Friday, saying that a recent steep drop in U.S. coronavirus cases and deaths “may be stalling” and “potentially leveling off at still a very high number” — a worrisome development that comes as more cases of concerning new variants have been found and could suggest that a return to normalcy is not yet quite as near as many Americans had hoped.
“Things are tenuous,” Rochelle Walensky, the director of the Centers for Disease Control and Prevention, said at a White House briefing on the pandemic. “Now is not the time to relax restrictions.”
Her warning was bolstered by Dr. Anthony S. Fauci, the government’s top epidemiologist, as the Biden administration scrambled to stay ahead of any new wave. President Biden himself flew to Houston to showcase the government’s latest mass vaccine site.
According to a New York Times database, virus cases across the United States appear to be leveling off from the steep decline that began in January, with figures comparable to those reported in late October. Cases have slightly increased week over week in recent days, though severe weather limited testing and reporting in Texas and other states the previous week, and not all states reported complete data on the Presidents Day holiday. The seven-day average of new cases was 77,800 as of Thursday.
While deaths tend to fluctuate more than cases and hospital admissions, Dr. Walensky said at the briefing on Friday, the most recent seven-day average is slightly higher than the average earlier in the week. The seven-day average of newly reported deaths was 2,165, as of Thursday.
“We at C.D.C. consider this a very concerning shift in the trajectory,” she said, adding, “I want to be clear: cases, hospital admissions and deaths — all remain very high and the recent shift in the pandemic must be taken extremely seriously.”
Dr. Walensky said some of the rise may be attributable to new variants of the coronavirus that spread more efficiently and quickly. The so-called B.1.1.7 variant, which first emerged in Britain, now accounts for approximately 10 percent of all cases in the United States, up from one to four percent a few weeks ago, she said. The U.S. ability to track variants is much less robust than Britain’s.
“I know people are tired; they want to get back to life, to normal,” she said. “But we’re not there yet.”
As cases had declined, some governors around the United States have begun to relax pandemic restrictions. States with Republican governors appeared to be more eager to make rollbacks, though New York, which has a Democrat as governor, has also been easing restrictions on a variety of activities.
On Friday, Gov. Henry McMaster of South Carolina, a Republican, announced that on Monday, restaurants would be able to serve alcohol past 11 p.m., and residents would not need to get approval from the state to hold events with 250 people or more. To try to limit the spread of the virus, the state last year ordered bars to stop serving alcohol after 11 p.m., which is three hours earlier than the late-night bar crowd was used to.
Brian Symmes, a spokesman for Mr. McMaster, said the governor “appreciates perspectives that differ from his own” but “respectfully disagrees” with Dr. Walensky’s assessment.
In Arkansas, Gov. Asa Hutchinson announced Friday that he’d be lifting restrictions around capacity limits for bars, restaurants, gyms and large venues, but extending the state’s emergency order and mask mandate until March 31. The current emergency order was set to expire February 27.
On Thursday, Gov. Greg Abbott of Texas said he was considering lifting a statewide mask mandate in place since July.
In Mississippi, Gov. Tate Reeves said he was also considering pulling back some restrictions, particularly mask mandates for people who have been fully vaccinated. As of Friday, 13 percent of the state’s population has received at least one shot, and 6.2 percent have received two, according to a Times database.
Dr. Fauci echoed Dr. Walensky’s warnings that more rollbacks at state or local levels would be unwise, noting that case levels remained at a “very precarious position.”
“We don’t want to be people always looking at the dark side of things, but you want to be realistic,” he said. “So we have to carefully look at what happens over the next week or so with those numbers before you start making the understandable need to relax on certain restrictions.”
In Oregon, Gov. Kate Brown extended the state’s emergency order until May 2. The state recorded a sharp decreases in daily cases, hospitalizations and deaths this week, but citing the new variants Ms. Brown said that “now is not the time to let up our guard.”
Eileen Sullivan Remy Tumin, Nicholas Bogel-Burroughs and Mitch Smith contributed reporting.
 A trial site for treatments from Regeneron and Eli Lilly in Mesa, Ariz., last summer.Credit…Adriana Zehbrauskas for The New York Times
A trial site for treatments from Regeneron and Eli Lilly in Mesa, Ariz., last summer.Credit…Adriana Zehbrauskas for The New York Times
The federal government has agreed to buy 100,000 doses of a recently authorized Covid-19 treatment from Eli Lilly, increasing the supply of such drugs for patients who are high risk of becoming seriously ill but are not yet hospitalized.
Under the deal, announced on Friday, the government will pay $210 million and Eli Lilly will ship out the doses by the end of March. The government has the option to buy 1.1 million more doses of the treatment through November, but how many of those doses ultimately get ordered will depend in part on the course of the pandemic in the United States.
The treatment is a cocktail of monoclonal antibodies combining the Eli Lilly drug known as bamlanivimab — which was authorized last November and is in use for high-risk Covid-19 patients — with a second drug known as etesevimab. The combination received emergency authorization earlier this month from the Food and Drug Administration. Both drugs consist of artificial copies of the antibodies that are naturally generated when a person’s immune system fights off an infection.
The U.S. government previously agreed to buy nearly 1.5 million doses of bamlanivimab alone. Eli Lilly has delivered more than 1 million doses already, with the remainder to be delivered by the end of March. More than 660,000 doses of bamlanivimab have been shipped out to states and other jurisdictions.
Eli Lilly’s new combination therapy could offer an advantage over bamlanivimab alone if worrisome coronavirus variants — particularly B.1.351, the one first identified in South Africa — take off in the United States. While bamlanivimab alone was found in a lab study to be powerless against the B.1.351 variant, preliminary data suggest that the combination therapy may be better able to fight off variants. That’s because so-called escape mutations in the variants that may enable them to avoid one antibody may not work against the second.
Another monoclonal antibody cocktail, made by Regeneron, is also authorized in the United States. Nearly 100,000 doses of that therapy have been shipped out.
Antibody treatments got a publicity boost last fall when they were given to Donald J. Trump when he was infected in the last months of his presidency, and to other high-profile Republicans, but they were surprisingly underused in many places in their first months of availability. Overwhelmed hospitals did not prioritize the treatments, which are cumbersome and must be given via intravenous infusions. Many patients and doctors did not know to ask for them or how to find them.
In December, the federal government’s early data collected from hospitals suggested that they had given only about 20 percent of their supply to patients. But that picture is changing. Eli Lilly has seen usage of bamlanivimab alone rise to around 40 percent nationwide, with uptake much higher in some places, Janelle Sabo, who leads Eli Lilly’s work on Covid-19 antibodies, said in an interview earlier this week.
 People over the age of 65 wait in line at a mobile Covid-19 vaccination site in the Chinatown neighborhood of Los Angeles.Credit…Philip Cheung for The New York Times
People over the age of 65 wait in line at a mobile Covid-19 vaccination site in the Chinatown neighborhood of Los Angeles.Credit…Philip Cheung for The New York Times
As more people in the United States learn of someone close to them who has received a Covid-19 vaccine, they are becoming more open to getting the vaccine themselves, according to the latest survey from the Kaiser Family Foundation, which has been monitoring attitudes since December.
Across most demographic groups, vaccine hesitancy, though still substantial, continues to abate. Fifty-five percent of adults now say they have either received one dose or will get it as soon as they can, up from 34 percent in December.
The so-called “wait and see” group is inching down, too, to 22 percent in February from 31 percent in January. But Black adults (34 percent) and Latino adults (26 percent) remain more inclined than white adults (18 percent) to take a wait-and-see approach.
About one in five Americans still refuses to get the shot, or said they would only do so if compelled by work or school.
Kaiser also took note of the political affiliation of its survey respondents. From December to February, the proportion of Democrats who had either gotten the shot or wanted to soared to 75 percent from 47 percent. Republicans remained more hesitant, but there was a notable increase in the amount who were vaccinated or hoped to be, to 41 percent from 28 percent.
Views of the vaccine were divided by age as well. People 65 and older, among the first demographic groups eligible for the shot, were among the most enthusiastic, an attitude that crossed racial lines. But while nearly half of older white people said they had already received at least one dose, only about one-third of older Black people said they had.
The survey also examined the most common reasons for hesitation. People were most concerned about side effects and also about the rapidity with which the vaccines were developed. They were also still gripped by misinformation, fearing they would have to pay for the vaccine (it is available at no cost) and that it would give them Covid (it does not).
The Centers for Disease Control and Prevention said on Friday about 47.2 million people in the U.S. have received at least one dose of a vaccine, including about 22.6 million people who have been fully vaccinated.
 Alisa Haushalter, the director of the Shelby County Health Department in Tennessee, in March. The county’s mayor said Ms. Haushalter had submitted her resignation on Friday.Credit…Mark Weber/Daily Memphian, via Associated Press
Alisa Haushalter, the director of the Shelby County Health Department in Tennessee, in March. The county’s mayor said Ms. Haushalter had submitted her resignation on Friday.Credit…Mark Weber/Daily Memphian, via Associated Press
The top health official in Memphis submitted her resignation after state health officials accused her department, which oversees the vaccine rollout in the state’s most populous county, of wasting thousands of doses, the mayor of Shelby County, Tenn., said on Friday.
The swift downfall of Alisa Haushalter, the Shelby County Health Department director, came hours after a news conference in which Dr. Lisa Piercey, Tennessee’s health commissioner, laid out a series of stunning accusations of mismanagement by Ms. Haushalter’s department, including episodes in which a volunteer may have stolen doses and another administered shots to two children.
State health officials have been investigating the department for at least a week after Ms. Haushalter first reported that vaccine doses had expired, Dr. Piercey said.
Investigators confirmed the expiration of 1,578 doses last week, but it was only this week that Shelby County health officials acknowledged that an additional 840 doses had expired on Feb. 15, bringing the total to more than 2,400.
And Dr. Piercey said that when she met with the mayor on Thursday, he said that he had heard a rumor about a volunteer possibly stealing vaccines on Feb. 3, but Dr. Piercey said the theft was never reported to any state or federal authorities. The state health department alerted the Federal Bureau of Investigation to the possible theft shortly after that conversation, she said. Dr. Piercey also said that a volunteer had wrongly vaccinated two children on Feb. 3.
The City of Memphis on Wednesday took over the responsibility of storing, transporting and allocating the vaccines.
 A senior receives the coronavirus vaccine in Quebec on Thursday.Credit…Christinne Muschi/Reuters
A senior receives the coronavirus vaccine in Quebec on Thursday.Credit…Christinne Muschi/Reuters
Canada’s drug regulator authorized the AstraZeneca vaccine on Friday as well as a version of it developed by the Serum Institute of India. The vaccine, which was developed with Oxford University, has not yet been authorized for use in the United States but has been key to Britain’s rapid vaccination rollout.
The addition of a third vaccine, in addition to the offerings from Moderna and Pfizer-BioNTech, may help Canada alleviate a growing dissatisfaction about the sluggish pace of vaccination in the country.
“Vaccines will keep arriving faster and faster,” Prime Minister Justin Trudeau told a news conference. “We now have a third safe and effective vaccine.”
Mr. Trudeau said that the government expects to receive, within days, about 500,000 doses out of an order of two million doses of the Serum Institute version of the vaccine, known as Covishield. About 20 million doses will start arriving from AstraZeneca in the spring.
The AstraZeneca vaccine, the first virus vector-based inoculation for Covid-19 authorized in Canada, has had some stumbles. In trials in South Africa, where a more contagious virus variant has become dominant, it did not appear to protect people from mild or moderate illness, which led the country to halt its use of the vaccine.
Drug approval officials with Health Canada said on Friday that while some studies suggested that the AstraZeneca vaccine is less effective overall than others, the sample sizes used were not large enough to yield a clear conclusion. But in Germany, many people regard it as “second-class” compared to the vaccine developed by the German company BioNTech and Pfizer, and are avoiding AstraZeneca’s.
 Downtown Chicago. Most scientists are optimistic that the worst of the pandemic is behind us.Credit…Lyndon French for The New York Times
Downtown Chicago. Most scientists are optimistic that the worst of the pandemic is behind us.Credit…Lyndon French for The New York Times
Across the United States and the world, the coronavirus seems to be loosening its stranglehold. The curve of cases, hospitalizations and deaths has yo-yoed before, but never has it plunged so steeply and so quickly.
Is this it, then? Is this the beginning of the end?
The road ahead is potholed with unknowns: how well vaccines prevent further spread of the virus, whether emerging variants remain susceptible enough to the vaccines and how quickly the world is immunized, so as to halt further evolution of the virus.
And the greatest ambiguity is human behavior. Will Americans desperate for a return to pre-pandemic lifestyles continue to wear masks and distance themselves from family and friends? How much longer can communities keep businesses, offices and schools closed?
Covid-19 deaths are unlikely to again rise quite as precipitously as in the past, and the worst may be behind us. But if Americans let down their guard too soon and if the variants spread in the United States as they have elsewhere, another spike in cases may well arrive in the coming weeks.
Buoyed by the shrinking rates over all, governors are lifting restrictions across the United States and are under enormous pressure to reopen completely.
“Everybody is tired, and everybody wants things to open up again,” said Ashleigh Tuite, an infectious disease modeler at the University of Toronto. “Bending to political pressure right now, when things are really headed in the right direction, is going to end up costing us in the long term.”
Looking ahead to late March or April, the majority of scientists interviewed by The Times predicted a fourth wave of infections. But they stressed that it is not an inevitable surge if precautions are maintained for a few more weeks.
“Just hang in there a little bit longer,” Dr. Tuite said. “There’s a lot of optimism and hope, but I think we need to be prepared for the fact that the next several months are likely to continue to be difficult.”
 President Biden is pushing hard for a $1.9 trillion economic rescue plan. Polls show that some Republican voters support the proposal, even if the party’s leaders do not.Credit…Doug Mills/The New York Times
President Biden is pushing hard for a $1.9 trillion economic rescue plan. Polls show that some Republican voters support the proposal, even if the party’s leaders do not.Credit…Doug Mills/The New York Times
In Washington, Republicans stand united in opposition to President Biden’s first major legislative proposal, a $1.9 trillion economic rescue plan that they have labeled a bloated, budget-busting “blue state bailout.”
But in rural Maine, Anthony McGill, a self-identified conservative Republican, describes the bill as something else entirely: “Most of it sounds like a good idea,” he said.
While Mr. McGill doesn’t agree with all the provisions, he supports the central thrust of the bill — another round of direct stimulus payments to nearly all Americans.
“There’s a lot of people that could use those checks. I don’t know about needing them, but we could all use them,” said Mr. McGill, 52, who voted for former President Donald J. Trump in November. “The debt is so far out of hand that it’s a fantasy number at this point. We might as well just blow it out till everything collapses.”
As Democrats prepare to vote as soon as Friday to pass the relief package in the House, Republican elected officials are struggling to overcome intraparty divides over whether to embrace the major pieces of the proposal — as well as to reconcile with the fact that many Republican voters support the plan. While Democrats are working swiftly to move their bill, Republicans are consumed by sideshows like false claims of voter fraud and what they call cancel culture, which are two major themes of the annual Conservative Political Action Conference, known as CPAC, starting on Friday in Orlando, Fla.
The lack of a unified Republican economic message reflects an unsettled party that is unable to agree on how to chart a path through a Democratic-controlled Washington. While congressional Republicans take a scattershot approach to try to undermine the legislation, mayors and governors in their party are pushing for the plan, saying their states and cities need the federal aid to keep police officers on their beats, reopen schools and help small businesses.
Polling shows a significant number of Republican voters agree: More than four in 10 Republicans back Mr. Biden’s aid package, according to polling from the online research firm SurveyMonkey for The New York Times. Over all, 72 percent of Americans said they supported the bill, a number that includes 97 percent of Democrats.
Global Roundup
 A woman receiving her first injection of the Pfizer-BioNTech Covid vaccine in Pontoise, in the northwestern suburbs of Paris on Feb. 5.Credit…Andrea Mantovani for The New York Times
A woman receiving her first injection of the Pfizer-BioNTech Covid vaccine in Pontoise, in the northwestern suburbs of Paris on Feb. 5.Credit…Andrea Mantovani for The New York Times
BRUSSELS — In vaccine-hungry, cash-rich Europe, the hunt for more doses has nations trading with each other, weighing purchases from Russia and China, and fielding offers from middlemen ranging from real to outright frauds.
Anger has been building over the European Union’s sluggish Covid vaccine rollout, which has left the E.U. member nations far behind several other wealthy countries, and many E.U. states are now looking beyond the bloc’s underwhelming joint purchasing strategy.
An immense black — or at least gray — market has arisen, with pitches from around the world at often exorbitant prices. Sellers have approached E.U. governments claiming to offer 460 million doses of vaccines, according to early results of an investigation by the bloc’s anti-fraud agency that were shared with The New York Times.
While they still plan to get vaccines from the bloc, some nations are also trying to negotiate directly with drug makers and eyeing the murky open market, where they are still unsure of the sellers and the products. Some have also agreed to swap vaccines with each other, deals some of them now have reason to regret.
The European Union last year was slow to make massive advance purchases from drug companies, acting weeks after the United States, Britain and a handful of other countries. This year, the bloc was blindsided by slower-than-expected vaccine production, and individual countries have fumbled the rollout.
About 5 percent of the E.U.’s nearly 450 million people have received at least one dose of a vaccine, versus almost 14 percent in the United States, 27 percent in Britain and 53 percent in Israel, as of earlier this week, according to the Our World in Data database and governments.
The stumbles by the world’s richest bloc of nations have turned vaccine politics toxic. Particularly galling to many Europeans is the sight of a former E.U. member, Britain, forging ahead with its vaccination and reopening plans, while many of their own societies remain under lockdown to contain a new surge of dangerous variants, their economies sinking deeper into recession.
In other international news on the pandemic:
-
The government in France has announced stricter border checks between Germany and the eastern Moselle region, one of several areas experiencing a spike in coronavirus infections. It is the latest pandemic measure to challenge the E.U.’s open borders. Officials said that starting Monday, those wanting to cross the border would have to present a negative coronavirus test from the past 72 hours. Cross-border workers in the area will be exempt.
-
President Emmerson Mnangagwa of Zimbabwe threatened this week to punish residents who do not take Covid vaccines. “You are not going to be forced to be vaccinated, but the time shall come when those who are not vaccinated won’t get jobs,” he said on Wednesday. The country of 15 million people received 200,000 donated doses from the Chinese company Sinopharm, and 600,000 more doses are expected in early March. Zimbabwe is also set to receive more than 1.1 million doses as part of the Covax distribution program for poor and middle-income countries.
-
North Korea’s borders have been closed for a year because of the pandemic, but some Russian diplomats and their families found a way home on a route that included a bus ride and a trip on a hand-pushed railroad trolley. Russia’s Foreign Ministry said on Friday that the journey by trolley was the only possible way for them to cross the border.
 Vaccines are administered at the Andrew Jackson Community Center in the Bronx this month. Credit…James Estrin/The New York Times
Vaccines are administered at the Andrew Jackson Community Center in the Bronx this month. Credit…James Estrin/The New York Times
Members of the Congressional Black Caucus and Dr. Anthony Fauci sought to reassure Black Americans that vaccine rollouts would be as equitable as possible and tried to quell anxieties over the safety of the inoculation among people of color during a televised forum Friday night.
Dr. Fauci, the government’s top infectious disease expert, said increasing pharmacies’ vaccine inventory and creating mobile units to get to hard-to-reach areas will help.
Experts say that Black and Latino Americans are being vaccinated at lower rates because they face obstacles like language barriers and inadequate access to digital technology, medical facilities and transportation. But mistrust in government officials and doctors also plays a role and is fed by social media misinformation. And in cities across the country, wealthy white residents are lining up to be vaccinated in low-income Latino and Black communities.
“It’s affecting us like no other disease, like no other epidemic has. That’s because for Black Americans, we were already behind,” Representative Barbara Lawrence, Democrat of Michigan, said during the forum on MSNBC’s “The Reid Out.”
“We’re looking at historic fear of vaccines and a fear of the health care industry because are they going to hurt us, harm us, ignore us or are they going to help us?” she said.
President Biden has said he wants to make racial equality a centerpiece of his vaccination rollout plan. He has begun shipping vaccines to federally qualified community health centers in Black and Latino communities.
While new data shows rates of vaccination in New York City’s Black and Latino communities are lower than rates in largely white communities and New York State’s Latino and Black residents were behind in vaccination totals, the absence of comprehensive national data on race and ethnicity makes it impossible to know just how equitable vaccine distribution is.
“This vaccine does not discriminate,” Ms. Lawrence said. “It hurts me when I see a line of people getting vaccines and my people are not represented but I’m invited to the funerals of those who have died. I have taken the vaccine. I believe in it. I need my people, my community, to step up.”
 A Covid-19 vaccination at Howard University in Washington, D.C., in December 2020.Credit…Tasos Katopodis/Getty Images
A Covid-19 vaccination at Howard University in Washington, D.C., in December 2020.Credit…Tasos Katopodis/Getty Images
The phone and online registration system set up by the District of Columbia for its residents to use to schedule Covid-19 vaccination appointments has been faltering after the city broadened the eligibility of prioritized residents who can make appointments on Thursday.
D.C. widened the criteria from people 65 and older to include residents with certain health conditions and those working jobs that require contact with multiple people.
But many complained on Thursday and Friday that the phone number listed for scheduling a jab was not in service, and that the website was returning error messages. Some people were able to secure an appointment, though it was not immediately clear why they were successful when others were not.
“In short … it did not go well,” one of the city’s council members, Charles Allen, said in an email to constituents on Thursday.
Mayor Muriel Bowser blamed the problems on the “high volume of traffic” on the city’s vaccination site and said in a tweet that the city was trying to fix the problems quickly. The mayor’s office did not immediately respond to a request for comment.
The city’s health department apologized for the problems and said the “technical review failure” on Thursday had been addressed. However, the problems persisted on Friday when the city released another 4,350 vaccine appointments.
As of Friday, nearly 11 percent of D.C. residents had received one dose of a vaccine, and nearly 5 percent had received two doses, according to a New York Times database.
 President Jair Bolsonaro of Brazil at his official office at the Planalto Palace in Brasilia on Wednesday. Credit…Ueslei Marcelino/Reuters
President Jair Bolsonaro of Brazil at his official office at the Planalto Palace in Brasilia on Wednesday. Credit…Ueslei Marcelino/Reuters
RIO DE JANEIRO — The daily death toll of Covid-19 in Brazil hit a record high of 1,582 on Thursday, according to a news consortium’s survey of local health departments. The painful milestone came as President Jair Bolsonaro railed against face masks, despite compelling scientific evidence that they are effective at preventing infections.
A rise in coronavirus infections in several states in Brazil, which officials say is being driven by more contagious variants, has overwhelmed hospitals across the country.
While epidemiologists and health officials warn of a worsening crisis in the weeks ahead, Mr. Bolsonaro took aim at masks during his weekly address on Facebook Thursday. Citing an unspecified German study, the president said masks were bad for children and that wearing them could lead to headaches, difficulty concentrating and a “decreased perception of happiness.”
Mr. Bolsonaro has been criticized at home and abroad for his cavalier response to the coronavirus pandemic. He has questioned the use of quarantine measures, social distancing and has sowed doubts about vaccines, saying he does not intend to get a shot.
While new cases and deaths are dropping in a number of other countries that were hard hit by the pandemic, including the United States, Brazil is in the grip of a second wave that began in November and shows no sign of easing.
The country began vaccinating medical professionals and older people last month, but the campaign is off to a slow start because the government has struggled to procure enough doses to quickly inoculate its 212 million citizens.
This week, Brazil’s Covid death toll surpassed 250,000, which is second only to the U.S. count of more than 500,000 deaths. The Brazilian health minister, Eduardo Pazuello, said on Thursday that the country had entered a “new stage of the pandemic” as a result of variants that he said are three times more contagious than earlier strains of the virus. “That is the reality we’re living today in Brazil,” he said.
 A doctor administering the Pfizer-BioNTech vaccine in Salisbury, England, last month.Credit…Neil Hall/EPA, via Shutterstock
A doctor administering the Pfizer-BioNTech vaccine in Salisbury, England, last month.Credit…Neil Hall/EPA, via Shutterstock
For people who have had Covid-19, a single dose of the Pfizer vaccine is enough to provide robust protection from the coronavirus, according to two new studies from Britain that were published late Thursday in The Lancet, a prominent medical journal.
The studies, among the first fully vetted papers to weigh in on how to vaccinate people who have had Covid-19, added strong evidence to the case for inoculating people who already have antibodies against the virus — but only with one dose of the Pfizer vaccine.
One of the studies, led by researchers at University College London and Public Health England, described the benefits of that strategy.
“This could potentially accelerate vaccine rollout,” they said. And that in turn could forestall dangerous new mutations: “Wider coverage without compromising vaccine-induced immunity could help reduce variant emergence,” the paper said.
In recent weeks, several studies on the topic were posted online that were not yet published in scientific journals, showing that one dose of a coronavirus vaccine amplified people’s antibodies from an earlier infection.
People’s immune responses to being infected are highly variable: Most people make considerable and long-lasting antibodies, while others who had milder infections produce relatively few, making it difficult to know how protected they are from the virus.
Vaccines act as a sort of booster for those people’s immune responses, inducing enough antibodies to offer protection. But a single dose, rather than the full two-dose protocol, is enough for those who have been infected, a number of studies have suggested.
Some researchers in the United States are trying to persuade the Centers for Disease Control and Prevention to recommend giving only one dose to people who have recovered from Covid-19. The studies from Britain seem likely to put pressure on health officials there to consider the same approach.
More than 28 million people in the United States and four million people in Britain, along with many others whose illnesses were probably never diagnosed, have been infected so far.
One of the new studies — led by Charlotte Manisty, a professor at University College London, and Ashley D. Otter, a research scientist at Public Health England — tracked 51 health workers in London who have submitted to routine tests for antibodies and infection since March. That gave researchers an unusually detailed picture of any pre-existing protection from the virus.
Roughly half of the health workers had experienced a mild or asymptomatic infection. And a single dose of the Pfizer vaccine increased their antibody levels more than 140-fold from their peak levels before being inoculated, the study said. That appeared to give them better protection against the coronavirus than two doses of the vaccine did in people who had never been infected, the researchers wrote.
The study raised the idea of giving people blood tests in the weeks before they became eligible for a Pfizer vaccine to determine whether they already had antibodies. People’s immune responses to an infection are highly variable, making it difficult to predict without a blood test who can be fully protected with a single dose.
As a further benefit of the single-dose strategy, the researchers wrote that it would spare people who have already been infected from the unpleasant side effects that sometimes follow a booster shot in that group.
The second study, led by scientists at Imperial College London, measured the immune responses of 72 health workers who were vaccinated in late December. A third showed signs of having previously been infected.
For those people, one dose of the Pfizer vaccine stimulated “very strong” antibody responses, the study said, as well as “very strong T-cell responses,” referring to another arm of the immune system.
It is not clear how long the post-vaccine immune response will last in people who have previously been infected compared with those who have not.
 Mark Machin, the chief executive of the Canada Pension Plan Investment Board, speaks at the Milken Institute Global Conference in Beverly Hills, Calif., in April 2019.Credit…Lucy Nicholson/Reuters
Mark Machin, the chief executive of the Canada Pension Plan Investment Board, speaks at the Milken Institute Global Conference in Beverly Hills, Calif., in April 2019.Credit…Lucy Nicholson/Reuters
The head of Canada’s largest pension fund stepped down on Thursday evening after news broke that he had flown to the United Arab Emirates to receive a coronavirus vaccine, despite federal rules banning inessential travel and a long line of older and immunocompromised citizens across the country waiting for their shots.
The Canada Pension Plan Investment Board announced in a news release on Friday that it had accepted the resignation of its chief executive, Mark Machin, who had “decided to travel personally to the United Arab Emirates where he arranged to be vaccinated against Covid-19.” The board added: “We take that responsibility of leadership very seriously.”
At about $375 billion, the fund is Canada’s largest, with more than 20 million contributors and beneficiaries.
Mr. Machin, who last year earned about $4.2 million at the helm of the crown corporation, is the latest public figure in the country to be publicly sanctioned for traveling abroad for personal reasons, while much of the country is hunkered down during the pandemic’s second wave.
His trip was perceived as not just selfish, but as queue-jumping by many Canadians, who have grown increasingly impatient with the sluggish rollout of vaccinations across the country. Less than 4 percent of the country’s 38 million people have received a dose — far fewer than most Western nations.
The Canada Pension Plan Investment Board is an independent corporation, but the country’s finance minister appoints its directors. Through her spokeswoman, the deputy prime minister and finance minister, Chrystia Freeland, called Mr. Machin’s decision “very troubling” and said Canadians expect the organization to “be held to a higher standard.”
Mr. Machin’s trip was first reported by The Wall Street Journal on Thursday night. Soon after, he sent an internal memo to staff stating the trip was supposed to be “very private” and that he was disappointed it has become the focus of “expected criticism,” The Globe and Mail reported.
 People walking in Charleston, S.C., at the end of January. A stubbornly high number of coronavirus cases farther away from the coast has kept the state’s case count relatively high in the last week.Credit…Cameron Pollack for The New York Times
People walking in Charleston, S.C., at the end of January. A stubbornly high number of coronavirus cases farther away from the coast has kept the state’s case count relatively high in the last week.Credit…Cameron Pollack for The New York Times
Even as the overall number of coronavirus cases in South Carolina declines, the situation away from the state’s coastline has remained stubbornly dangerous, with some counties in the central and western parts of the state reporting some of the most dire case counts relative to their populations in the Southeast.
Those regions have kept the state’s case count from declining further, and over the last week, South Carolina has reported more cases per capita than any other state, as of Thursday.
The cases are staying high in a mix of small and large counties, including the metropolitan areas of Columbia, the state’s capital and second-largest city, and Spartanburg. Those two metro areas have reported more cases per capita over the last two weeks than all but three other metro areas in the United States, according to a New York Times database. And they are doing so even as cases in Charleston, the state’s largest city, have consistently fallen.
Gov. Henry McMaster cited the overall decline in U.S. cases on Friday in deciding to roll back several public health measures beginning next week. Starting Monday, there will no longer be an 11 p.m. curfew for alcohol sales at restaurants, and large gatherings can resume, though he encouraged people to voluntarily follow the guidelines that had been in place.
“With the spread of the virus consistently decreasing across the country and more of the most vulnerable South Carolinians being vaccinated every day, I believe these targeted and limited safety measures are no longer necessary,” Mr. McMaster said in a statement. “The virus is still among us and we all must continue to make responsible decisions to take care of ourselves and our loved ones, but those decisions are for South Carolinians to make.”
The move from Mr. McMaster came on the same day that Dr. Rochelle Walensky, the director of the Centers for Disease Control and Prevention, warned that the recent decline in cases may be stalling and said governors should keep public health measures in place. “Now is not the time to relax restrictions,” she said.
Brian Symmes, a spokesman for Mr. McMaster, said the governor “appreciates perspectives that differ from his own” but “respectfully disagrees” with Dr. Walensky’s assessment.
One day earlier, on Thursday, South Carolina’s top epidemiologist, Dr. Linda Bell, said in an interview with a local television station that while it was understandable that residents would be optimistic as vaccines rolled out, it was important for them to not forget that the situation was still severe in much of the state.
“We cannot say yet that we have passed the worst part of the pandemic,” Dr. Bell said on WIS-TV.
“I don’t want people to be too encouraged to the point that they stop doing the preventive measures, looking at the current decline in cases,” she added, “because we have declined to a level that has taken us to the height of cases that we were seeing after the Fourth of July, and I want to remind people that we were actually very alarmed.”
Mitch Smith contributed reporting.
 Members of the Kansas State University marching band maintained social distance as they played before a college football game in October 2020.Credit…Charlie Riedel/Associated Press
Members of the Kansas State University marching band maintained social distance as they played before a college football game in October 2020.Credit…Charlie Riedel/Associated Press
Colleges and universities across the country are pledging to reopen more fully in the fall, with some administrators worried that students won’t return to campus if normality, or some semblance of it, isn’t restored by September.
Schools from large state institutions to small private ones have announced they are laying plans to bring students back to dormitories, deploy professors to teach most (if not all) classes in person and restart extracurricular activities, in stark contrast to the past academic year of largely virtual courses and limited social contact. The announcements of these changes coincide with the sending of acceptance letters to the class of 2025.
Some schools have taken a financial hit because of deferred admissions or lost room-and-board fees.
Bradley University, in Peoria, Ill., which has 5,600 undergraduate and graduate students, said earlier this month that it would return to “traditional residential education” in the fall, with in-person classes and activities on campus.
Kansas State University announced on Wednesday that it too is planning a “more normal” fall semester, with largely in-person classes, events and activities. Ohio State announced on Thursday that it plans to offer “robust” in-person activities and classes, allowing students to live in residence halls and fans to attend football games.
Katherine Fleming, New York University’s provost, told colleagues in an email on Tuesday of plans to have “all faculty teaching their classes in-person, in the classroom, in the fall 2021.” She conceded, however, that this would depend in part on whether enough professors were vaccinated by then.
Indeed, most school officials said that whether they can deliver on these promises hinges on factors like how much the virus can be suppressed, the availability of the vaccine — which is still in scarce supply, even for those who are eligible — and guidance from government authorities.
Despite their hopefulness about the fall, schools have struggled with keeping the virus in check. Positivity rates rose among college students, as among the general population, over the holidays, when people traveled. Administrators have put out many stern warnings that small parties and gatherings have been a source of infection. Many have noted, however, that the classroom itself has not proven to be a vector of infection, as long as students and teachers follow safety guidelines like wearing masks and social distancing.
More than 120,000 coronavirus cases have been linked to American colleges and universities since Jan. 1, and more than 530,000 cases have been reported since the beginning of the pandemic, according to a New York Times survey. The Times has identified more than 100 deaths, but the vast majority involved employees, not students.
Video
transcript
Back
transcript
Biden Tours Texas Emergency Aid Centers After Winter Storms
President Biden spent the day in Houston touring a local food bank and other areas hit by recent snowstorms. Storm damage is expected to total $20 billion, according to the Insurance Council of Texas.
Mr. President, welcome. This is our emergency operations center, Harris County Emergency Operations Center. For me and these folks you see here, this has been our home away from home. Over fire, flood, Covid and now this winter weather event. These folks are the tip of the sphere. Thank you for What you are doing. All I did is I got a call and before you asked the question I just said yes. He did. He said yes before I even asked the question. Absolutely. It’s incredible. It’s an incredible place. They’re doing — and they have so many talented people here, not just volunteers, really talented people.
 President Biden spent the day in Houston touring a local food bank and other areas hit by recent snowstorms. Storm damage is expected to total $20 billion, according to the Insurance Council of Texas.CreditCredit…Doug Mills/The New York Times
President Biden spent the day in Houston touring a local food bank and other areas hit by recent snowstorms. Storm damage is expected to total $20 billion, according to the Insurance Council of Texas.CreditCredit…Doug Mills/The New York Times
At an emergency response center in Houston, President Biden praised officials who’d slept in stairwells as they worked around the clock to answer the call of terrified residents who had no power or drinkable water when devastating snowstorms hit. At a food bank, Mr. Biden hugged a little girl who was volunteering. He then turned to a woman to discuss the death of his eldest son, plugging into the pain of people around him by accessing his own.
“Incredible,” Mr. Biden said as he surveyed what was happening around him. “It’s absolutely amazing, and we could do so much more.”
Infrastructure and coronavirus relief may be on the official agenda in Washington, but the overwhelming nature of grief was the unofficial theme of the week.
On Monday, Mr. Biden presided over a solemn observance of a grim pandemic milestone: more than 500,000 Americans dead. “While we have been fighting this pandemic for so long, we have to resist becoming numb to the sorrow,” he said during a speech at that White House that evening.
And when he traveled to Houston with Jill Biden, the first lady, the president for the first time used his new platform to show support for a community ravaged by twin crises of devastating snowstorms and the pandemic.
“You’re saving people’s lives,” Mr. Biden told a group of officials at an emergency operations center. “As my mother would say, you’re doing God’s work.”
Across Texas, the damage from the storms is extensive and the recovery is expected to be slow. The damage is expected to cost upward of $20 billion, according to the Insurance Council of Texas. Coronavirus vaccinations had all but stalled because of the storm, but are beginning to rebound — Mr. Biden was expected to visit a mass-vaccination site at a nearby stadium later Friday.
As soon as Mr. Biden hit the ground, the tone of his visit was different from what victims of natural disasters encountered when his predecessor, Donald J. Trump, would visit. Mr. Trump more than once threatened to withhold disaster funding if he had toxic political relationships with officials in those places.
Mr. Biden, who has been pushing a $1.9 trillion coronavirus relief package, was joined by Republicans who praised him for approving a major disaster declaration for Texas, ensuring the flow of federal resources to some 126 counties across the state that were hit by the storms — about half the number requested by Gov. Greg Abbott, who joined Mr. Biden on the trip.
“The governor and Senator Cruz and I asked for a declaration from the federal government which provides access to public and private assistance through FEMA,” said Senator John Cornyn, Republican of Texas and another participant on the trip, referring to the state’s junior senator, Ted Cruz. “That’s going to be important for our recovery.”
Mr. Cruz did not participate. He was in Florida, speaking at the Conservative Political Action Conference.
In Harris County, where Houston is, about 50 percent of 4.9 million residents lost power as the storms hit. Nearly two weeks later, about 10,000 residents are still boiling their water, according to county officials. More than 50,000 across the state still do not have access to safe water, according to officials with the Federal Emergency Management Agency.
During the visit to the food bank, Dr. Biden slipped cans of peaches into packages of food for students who rely on free school meals. Mr. Biden talked to children and told them about his own family.
It was a marked difference from Mr. Trump, who was criticized in 2018 for visiting a disaster relief center in Puerto Rico, only to throw paper towels at people who’d survived a Category 5 hurricane. “I was having fun,” Mr. Trump said afterward. “They were having fun.”