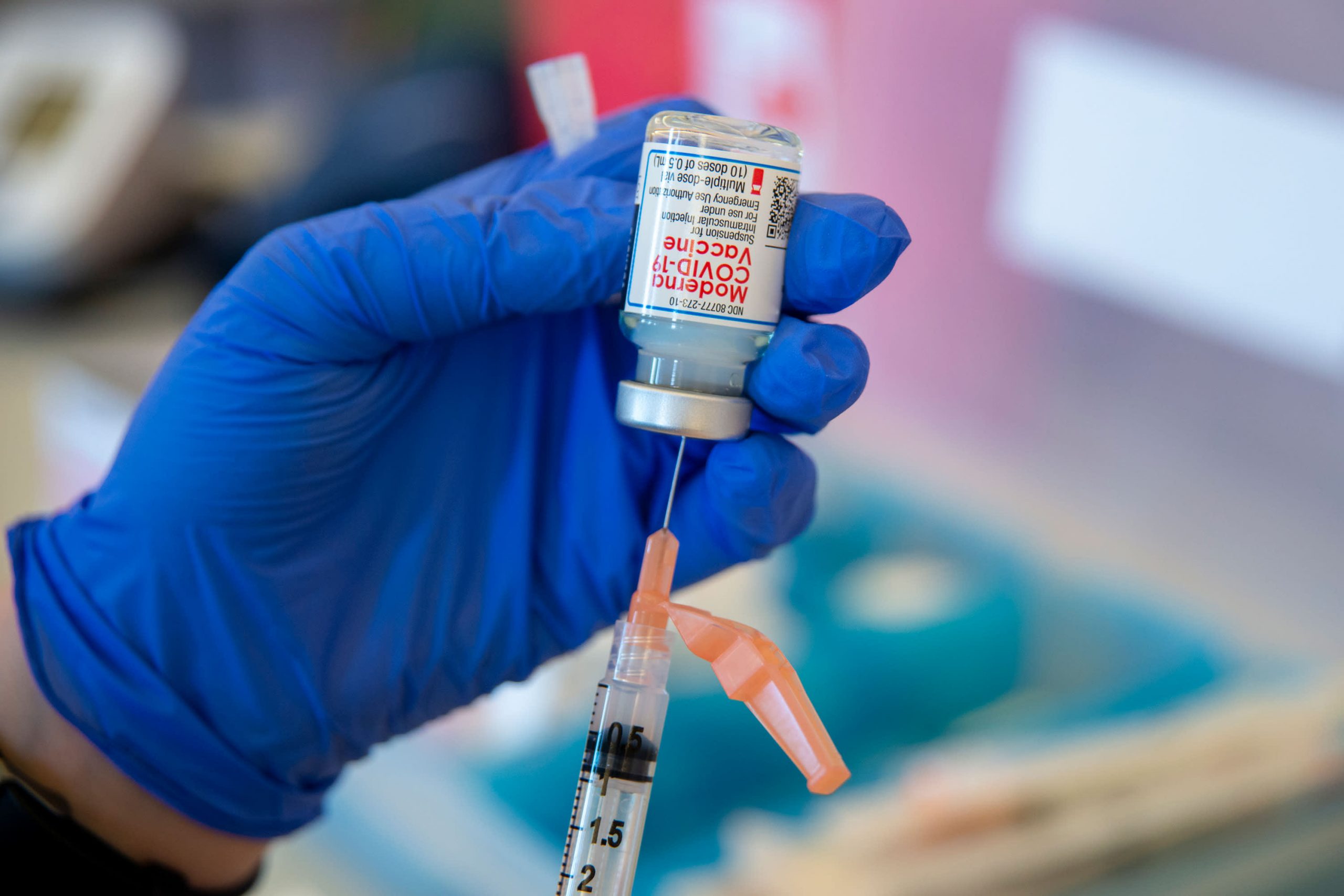
Moderna is asking U.S. regulators to agree to what could be a remarkably simple proposal to speed up Americans’ immunization against the coronavirus: fill the empty space in the vials with up to 50 percent more doses.
The Food and Drug Administration could decide in a matter of weeks whether Moderna, a Massachusetts-based biotech company that develops one of two state-approved Covid vaccines, can increase the number of doses in its vials from up to 15. Moderna has ramped up production of its vaccine, but the process of filling, capping and labeling millions of tiny vials has proven to be a roadblock. The company could produce more if regulators allow it to make fuller vials, Ray Jordan, a Moderna spokesman, said late Monday.
While it’s not clear how quickly Moderna could adjust its production, any spike could be extremely welcome news in the campaign to contain a pandemic that has killed more than 443,000 people in the US alone.
“That just makes a lot of sense,” said Dr. Nicole Lurie, who was Assistant Secretary of Health for Preparedness and Response under President Barack Obama. If Moderna can use the same size vial and the same production lines that are already running, she said, “It’s a relatively simple and straightforward solution.”
Moderna has given up about 46 percent of the vaccine previously administered in the United States, according to the Centers for Disease Control and Prevention. The remainder comes from Pfizer-BioNTech, the only other vaccine developer to receive emergency clearance.
Both vaccines require two doses, and both companies have promised to deliver 200 million doses by July. That would be enough to cover roughly three-quarters of the nation’s adults. If Moderna can deliver cans faster, that schedule could accelerate.
The Biden administration is looking for a way to strengthen production, for example for obstacles in the “filling and finishing” phase of production. Although this stage of the nuts and bolts receives less attention than the manufacturing process of the vaccine itself, it has been identified as a production limitation for years.
Moderna has discussed the possible change with the Food and Drug Administration but has not yet provided manufacturing data for support, according to those familiar with the discussions. Federal regulators may be receptive to the idea of more doses in each vial, but could prevent a 50 percent increase and instead approve a more modest number of additional doses.
The industry standard has long been 10 doses per vial, and federal regulators may fear that having too many extra punctures by needling the rubber cover of the vial and the time it takes to extract more doses increases the risk of contamination of the vaccine with Bacteria could increase.
At some point, too much liquid can cause a vial to break. Moderna has tested what happens when additional doses are added and found the limit to be 15, according to people familiar with the company’s operations and not authorized to speak publicly.
Moderna’s proposal for a five dose increase was previously reported by CNBC.
Packing more vaccines in each vial is one of several options the White House and health officials are considering to ramp up production before spring, when authorities expect a renewed spike in infections from emerging variants of the coronavirus. Some ideas, such as combining fractions of doses left over in vials, have been suggested and discarded.
Pfizer is unable to increase the amount of vaccine in its vials because its manufacturing is geared towards a specific vial size that can only hold about six doses. Moderna’s vial is large enough to hold more than the 10 doses currently allowed, so it can add more without creating a new production line.
When asked about Moderna’s proposal, a White House spokesman said Monday that “all options are on the table”.
Prashant Yadav, who studies healthcare supply chains at the Washington Center for Global Development, said Moderna could potentially make more of its vaccine “relatively quickly” if it were given the green light to add doses to each vial.
Covid19 vaccinations>
Answers to your vaccine questions
Am I eligible for the Covid vaccine in my state?
Currently more than 150 million people – almost half of the population – can be vaccinated. But each state makes the final decision on who goes first. The country’s 21 million healthcare workers and three million long-term care residents were the first to qualify. In mid-January, federal officials asked all states to open eligibility to anyone over 65 and adults of any age with medical conditions that are at high risk of becoming seriously ill or dying of Covid-19. Adults in the general population are at the end of the line. If federal and state health authorities can remove bottlenecks in the distribution of vaccines, everyone over the age of 16 is eligible as early as spring or early summer. The vaccine has not been approved in children, although studies are ongoing. It can take months before a vaccine is available to anyone under the age of 16. For the latest information on vaccination guidelines in your area, see your state health website
Is the Vaccine Free?
You shouldn’t have to pay anything out of pocket to get the vaccine, despite being asked for insurance information. If you don’t have insurance, you should still get the vaccine for free. Congress passed law this spring banning insurers from applying cost-sharing such as a co-payment or deductible. It consisted of additional safeguards prohibiting pharmacies, doctors, and hospitals from charging patients, including uninsured patients. Even so, health experts fear that patients will end up in loopholes that make them prone to surprise bills. This may be the case for people who are charged a doctor’s visit fee with their vaccine, or for Americans who have certain types of health insurance that are not covered by the new regulations. When you get your vaccine from a doctor’s office or emergency clinic, talk to them about possible hidden costs. To make sure you don’t get a surprise invoice, it is best to get your vaccine at a Department of Health vaccination center or local pharmacy as soon as the shots become more widely available.
Can I choose which vaccine to get?How long does the vaccine last? Do I need another next year?
That is to be determined. It is possible that Covid-19 vaccinations will become an annual event just like the flu vaccination. Or the vaccine may last longer than a year. We’ll have to wait and see how durable the protection from the vaccines is. To determine this, researchers will track down vaccinated people to look for “breakthrough cases” – those people who get Covid-19 despite being vaccinated. This is a sign of a weakening of protection and gives researchers an indication of how long the vaccine will last. They will also monitor the levels of antibodies and T cells in the blood of people who have been vaccinated to see if and when a booster shot might be needed. It is conceivable that people might need boosters every few months, once a year, or just every few years. It’s just a matter of waiting for the data.
Does my employer need vaccinations?Where can I find out more?
But he said it wasn’t an instant change. “I don’t think Moderna has a surplus,” he said.
Dr. Lurie, an advisor to the Coalition for Epidemic Preparedness Innovations, known as CEPI, said that during the federal government’s H1N1 swine flu response, the problem of filling and completion created a “major bottleneck” with a limited supply of vaccines.
She said that at the start of the coronavirus pandemic at CEPI, there was discussion about which vial size would be suitable for mass vaccination: five, 10 or 20. Last year, a global shortage of glass vials became apparent due to negotiations between the federal government and vaccine manufacturers , which added further stress to drug companies developing coronavirus vaccines.
Mr Yadav said the finish-and-fill process has been automated to prevent contamination and ensure precise dosing of the microgram. It can fill up to 1,000 vaccine bottles per minute at top speed, he said.
A 15-dose vial made a compromise, he said. There could be more waste if healthcare professionals run out of people to be vaccinated and have to discard the remaining doses. But during a raging pandemic, experts said that could be a risk federal health officials would take.
Dr. Moncef Slaoui, who led the Trump administration’s vaccine development program and is an advisor to the Biden administration through next week, said other big drug companies like Merck or GlaxoSmithKline could potentially be able to end some of the fill and leg burden.
“It’s a more general type of manufacturing activity,” he said.
French drug maker Sanofi announced last week that it would produce more than 100 million doses of the Pfizer BioNTech vaccine starting this summer to meet demand in Europe. Company employees said Sanofi will fill and package vials at a Sanofi facility in Frankfurt, near BioNTech’s German headquarters. BioNTech, Pfizer’s German partner, developed the vaccine.