Here’s what you need to know:
Credit…Atul Loke for The New York Times
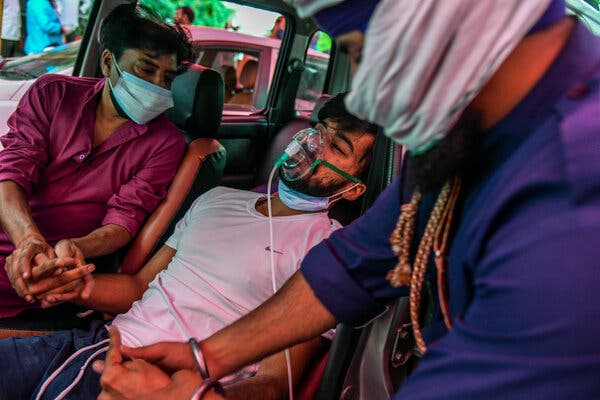
As a second wave of the pandemic rages in India, which set a global record new cases for the fifth consecutive day on Monday, countries around the world are trying to help. But their efforts to send oxygen and other critical aid are unlikely to plug enough holes in India’s sinking health care system to end its deadly catastrophe.
The Indian health ministry reported almost 353,000 new cases and 2,812 deaths on Monday. Enormous funeral pyres have spilled into parking lots and city parks. Experts say that India’s reported overall toll of more than 195,000 deaths could be a vast undercount.
The emergency in India, where a worrying virus variant is spreading rapidly, has global implications for potential infections worldwide, as well as for countries relying on India for the AstraZeneca vaccine, millions of doses of which are manufactured there.
“It’s a desperate situation out there,” said Ramanan Laxminarayan, the founder and director of the Center for Disease Dynamics, Economics & Policy, adding that donations will be welcome, but may make only a “dent on the problem.”
Scientists fear that part of the problem is the emergence of a virus variant known as the “double mutant,” B.1.617, because it contains genetic mutations found in two other difficult-to-control versions of the coronavirus. One of the mutations is present in the highly contagious variant that ripped through California earlier this year. The other mutation is similar to one found in the variant dominant in South Africa and is believed to make the virus more resistant to vaccines.
Still, scientists caution it is too early to know with certainty how pernicious the new variant emerging in India really is.
In the early months of 2021, the government of Prime Minister Narendra Modi acted as if the coronavirus battle had been won, holding huge campaign rallies and permitting thousands to gather for a Hindu religious festival.
Now, Mr. Modi is striking a far more sober tone. He said in a nationwide radio address on Sunday that India has been “shaken” by a “storm.” And countries, companies and powerful members of the diaspora have pledged to pitch in.
Patients are suffocating in the capital, New Delhi, and other cities because hospitals’ oxygen supplies have run out. Frantic relatives have appealed on social media for leads on intensive-care-unit beds and experimental drugs. The government has extended New Delhi’s lockdown by another week.
India’s Supreme Court last week ordered the government to come up with a “national plan” for distributing oxygen supplies.
The problems in India’s hospitals go beyond oxygen shortages. In the western state of Gujarat, more than a dozen patients were evacuated from a hospital on Sunday night after an air-conditioning unit caught fire, the Press Trust of India reported, the third accident involving virus patients in India in the past seven days.
Mr. Modi appears to be looking to the rest of the world to help India quell the wave. Saudi Arabia and the United Arab Emirates have promised oxygen generators. The United States has pledged raw material for coronavirus vaccines and intends to share up to 60 million doses of the AstraZeneca vaccine with other nations, so long as the doses clear a safety review conducted by the Food and Drug Administration, officials said Monday. Indian-American businessmen have pledged millions in cash from the companies they lead.
At a news conference on Monday, Dr. Tedros Adhanom Ghebreyesus, the director general of the World Health Organization, called the situation in India “beyond heartbreaking.” He said the organization has deployed 2,600 staff to India to provide surveillance and vaccination help.
A global coronavirus surge, driven largely by the devastation in India, continues to break daily records and run rampant in much of the world, even as vaccinations ramp up in wealthy countries. More than one billion shots have now been administered globally.
On Sunday, the world’s seven-day average of new cases hit 774,404, according to a New York Times database, higher than the peak average during the last global surge, in January. Despite the number of shots given around the world, far too small a percentage of the global population of nearly eight billion have been vaccinated to slow the virus’s steady spread.
| United States › | United StatesOn Apr. 25 | 14-day change | |
|---|---|---|---|
| New cases | 33,662 | –16% | |
| New deaths | 282 | –3% | |
| World › | WorldOn Apr. 25 | 14-day change | |
|---|---|---|---|
| New cases | 378,263 | +15% | |
| New deaths | 7,655 | +4% | |
U.S. vaccinations ›
![]()
 Credit…Atul Loke for The New York Times
Credit…Atul Loke for The New York Times
President Biden, under intense pressure to do more to address the surging pandemic abroad, including a humanitarian crisis in India, intends to make up to 60 million doses of the AstraZeneca vaccine available to other countries, so long as federal regulators deem the doses safe, officials said Monday.
The announcement came after Mr. Biden spoke with Prime Minister Narendra Modi of India and the two pledged to “work closely together in the fight against Covid-19.” It is a significant, albeit limited, shift for the White House, which has until now been reluctant to make excess doses of coronavirus vaccine available in large amounts.
But the commitment is a tricky one to make: The AstraZeneca doses are manufactured at the Baltimore plant owned by Emergent BioSolutions, where production has been halted amid fears of contamination. The New York Times has reported extensively on problems at the plant, which had to throw out millions of doses of AstraZeneca vaccine between October and January, and later discarded up to 15 million doses of the vaccine developed by Johnson & Johnson, also because of concern about possible contamination.
AstraZeneca’s vaccine, unlike those of Pfizer, Moderna and Johnson & Johnson, has also not been granted emergency use authorization by the Food and Drug Administration. And the administration would not specify which countries will receive the vaccine.
Jen Psaki, the White House press secretary, cautioned at a news conference that the donations of doses would not happen right away. She said about 10 million doses could be released “in the coming weeks” if the F.D.A. determines that the vaccine meets “our own bar and our own guidelines,” and that another 50 million doses are in various stages of production.
“Right now we have zero doses available of AstraZeneca,” Ms. Psaki said.
In a statement, a spokesperson for AstraZeneca said that the company would not comment on specifics but that “the doses are part of AstraZeneca’s supply commitments to the U.S. government. Decisions to send U.S. supply to other countries are made by the U.S. government.”
Correction: April 26, 2021
An earlier version of this article referred incorrectly to a safety review that the Food and Drug Administration is required to conduct before AstraZeneca coronavirus vaccine doses are shared with other nations. The doses themselves must clear an F.D.A. safety review, not the plant where the doses are manufactured.
 Credit…Emile Ducke for The New York Times
Credit…Emile Ducke for The New York Times
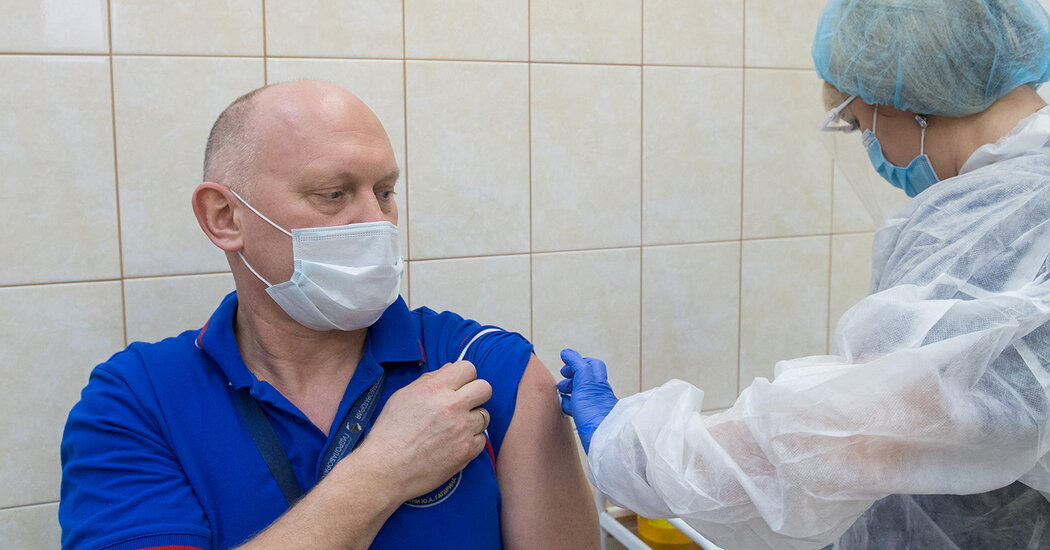
Brazil’s health authority, Anvisa, said late on Monday that it would not recommend importing Sputnik V, the Covid-19 vaccine developed by Russia.
Anvisa said that important safety tests had not been performed, and that questions remained about the vaccine’s development, safety and manufacturing.
Data about the vaccine’s efficacy were “uncertain,” Gustavo Mendes Lima Santos, Anvisa’s manager of medicine and biological products, said in a lengthy presentation explaining the health authority’s decision.
A tweet from the official Sputnik V Twitter account — in Portuguese — pushed back on Monday, saying that the vaccine’s developers had shared “all the necessary information and documentation” with Anvisa. In another tweet, it urged Anvisa that “we have no time to waste — let us start saving lives in Brazil. Together.”
Russia is using Sputnik V in its mass vaccination campaign, and the vaccine has been approved for emergency use in dozens of other countries. Its rollout has been entangled in politics and propaganda, with President Vladimir V. Putin announcing its approval for use even before late-stage trials began. For months, it was pilloried by Western scientists.
The Gamaleya Research Institute, part of Russia’s Ministry of Health, developed the vaccine, also known as Gam-Covid-Vac. A peer-reviewed study published in The Lancet in February said the vaccine had an efficacy rate of 91.6 percent.
Skepticism from Western experts has focused mostly on its early approval, not the vaccine’s design, which grew out of decades of research on adenovirus-based vaccines. Other Covid-19 vaccines are also based on adenoviruses, such as one from Johnson & Johnson using Ad26, and one by the University of Oxford and AstraZeneca using a chimpanzee adenovirus.
While Sputnik V’s developers have yet to release detailed data on adverse events observed during the trials, the Russian government has been using the vaccine to inoculate its own citizens for months. Russia has also exported Sputnik V to Belarus, Argentina and other countries, suggesting that any harmful side effects overlooked during trials would by now have come to light.
As vaccine supply woes in Europe worsened, the European Union’s drug regulator announced last month that it was reviewing the Sputnik V vaccine after member nations began announcing they would acquire the shot on their own.
Video
transcript
Back
transcript
E.U. Sues AstraZeneca
The European Union has sued AstraZeneca over its failure to deliver hundreds of millions of Covid vaccination doses by the end of June as promised.
Indeed, the commission has started last Friday a legal action against the company AstraZeneca on the basis of breaches of the advance purchase agreement. The reason indeed being that the terms of the contract or some terms of the contract have not been respected, and the company has not been in a position to come up with a reliable strategy to ensure the timely delivery of those. What matters to us in this case is that we want to make sure that there’s a speedy delivery of a sufficient number of doses that the European citizens are entitled to and which have been promised on the basis of the contract. So the commission has indeed started legal action on its own behalf and on behalf of the 27 member states that are fully aligned in their support for this procedure.
 The European Union has sued AstraZeneca over its failure to deliver hundreds of millions of Covid vaccination doses by the end of June as promised.CreditCredit…Alessandro Grassani for The New York Times
The European Union has sued AstraZeneca over its failure to deliver hundreds of millions of Covid vaccination doses by the end of June as promised.CreditCredit…Alessandro Grassani for The New York Times
The European Union has sued AstraZeneca over what the bloc has described as delays in shipping hundreds of millions of doses of coronavirus vaccines, a sharp escalation of a longstanding dispute between the bloc and the maker of one of the world’s most important vaccines.
AstraZeneca has said that it would be able to deliver only a third of the 300 million doses that European officials had been expecting by the end of June. As a result, European officials said on Monday that they believed AstraZeneca had broken its contract, and that they were seeking speedier deliveries than the company said it could muster.
The two sides’ relationship had grown acrimonious in January when AstraZeneca slashed its expected deliveries for the first quarter of the year, setting back the bloc’s vaccination campaign by weeks as cases picked up across the continent and political leaders faced scorching criticism for inadequate planning.
For AstraZeneca, whose cheap and easy-to-store shot is being used by 135 countries, the lawsuit could create further difficulties in a bruising stretch. No company had been as instrumental in the race to vaccinate poorer countries around the world, but AstraZeneca has been buffeted in recent weeks by the discovery of an exceedingly rare, though serious, side effect that has prompted restrictions on its use in parts of Europe.
At issue in the legal dispute was whether AstraZeneca had done everything in its power to meet its delivery schedule. Pascal Soriot, the company’s chief executive, has said that the contract required only that it make its “best efforts” to deliver the purchased doses on time.
Vaccine production is a notoriously fickle science, with live cultures needing time to grow inside bioreactors, for instance. In an effort to supply doses not only to richer nations that had purchased them well in advance, but also to poorer nations, AstraZeneca had partnered with manufacturing sites around the world, rather than relying on only a few factories, as Pfizer and Moderna have.
AstraZeneca, which developed the vaccine with the University of Oxford, has also said that the European Commission, the bloc’s executive branch, finalized its contract months after Britain did, giving the company less time to iron out any manufacturing difficulties.
Legal experts said that the “best efforts” language in the contract raised the burden on the Europeans to prove that AstraZeneca did not act diligently enough to supply the promised doses. But they also said that it did not entirely insulate the company from being deemed in breach of contract.
GLOBAL ROUNDUP
 Credit…Chris Mcgrath/Getty Images
Credit…Chris Mcgrath/Getty Images
President Recep Tayyip Erdogan of Turkey ordered a national lockdown for three weeks, closing nonessential businesses and sending all students home, as the nation struggles to contain the latest surge in cases of the coronavirus.
Turkey ranks fourth in the world in new daily cases per person, averaging 63 cases per 100,000 people, according to a New York Times database. Its seven-day average for deaths ranks 11th in the world.
The lockdown starts on April 29 and will end on May 17, coinciding with Eid al-Fitr, the end of the holy month of Ramadan, Mr. Erdogan said after meeting with his cabinet. Schools and restaurants will close and travel within Turkey will require a permit, he said. Government employees will either work from home or in shifts. Essential businesses like those in the food, manufacturing and health sectors will be exempt, Mr. Erdogan said.
“In a period where Europe is opening up, we have to pull the number of cases’’ lower, Mr. Erdogan said. “Otherwise, it would be inevitable to face a heavy cost from tourism to trade to education.’’
So far, about 16 percent of its total population has received at least one dose of the vaccine from Sinovac or Pfizer-BioNTech, according to data from the Our World in Data project at the University of Oxford.
Turkey reported about 63,000 new cases on April 16, its highest daily tally since the start of the pandemic.
In other updates from around the world:
-
Brazil’s health regulator rejected the use of Russia’s Sputnik V vaccine late on Monday, citing “inherent risks” and a lack of information about the vaccine’s safety and quality, Reuters reported.
-
The governments of Singapore and Hong Kong said on Monday that a long-delayed travel bubble between the two Asian financial centers would begin next month, allowing travelers on designated flights to bypass quarantine. The travel arrangement, which was originally supposed to begin last November, was suspended at the last minute when Hong Kong experienced a sudden surge in cases.
-
The Philippines surpassed the one million mark on Monday in the total number of coronavirus cases it has reported, as the country struggles with newer, deadlier forms of the virus. The Philippines reported very few cases last year, and did not see a significant surge until recently. In response, Manila and four other suburbs went into lockdown earlier this month.
-
For the first time in nearly nine months, Portugal’s health authority on Monday reported no coronavirus-related deaths in the last 24 hours, according to Reuters. Portugal has reported nearly 17,000 Covid-19 deaths and more than 830,000 cases.
-
Health authorities in Germany will allow all adults to sign up for vaccine appointments beginning in June, Chancellor Angela Merkel said on Monday. The announcement came after a meeting with lawmakers to discuss lifting social restrictions for fully vaccinated people, a sign that Germany may be moving closer to emerging from its latest lockdown.
-
More than 78,000 people attended an Australian rules football match in Melbourne on Sunday night in what is believed to be the world’s biggest crowd at a sporting event since the coronavirus pandemic began. Just three days earlier, the government of the state of Victoria, of which Melbourne is the capital, had increased the attendance cap for the 100,000-capacity venue, the Melbourne Cricket Ground, to 85 percent from 75 percent.
 Credit…Andy Buchanan/Agence France-Presse — Getty Images
Credit…Andy Buchanan/Agence France-Presse — Getty Images
Scotland and Wales reopened restaurants, cafes, and nonessential shops on Monday, marking the next phase of a gradual relaxation of coronavirus restrictions that have been in place for months.
In Scotland, restaurants can serve food but not alcohol indoors until 8 p.m., and they can serve food and alcohol outdoors without restrictions. Stores, beauty salons, museums and galleries also reopened, and people are permitted to book travel in the rest of Britain.
The first minister of Scotland, Nicola Sturgeon, said she was hopeful that the country would continue its progress and lift more restrictions by the summer. But she cautioned that the virus was more infectious now than it had been in earlier waves and, therefore, “We must stick to the rules.” Free rapid tests will be available to the public.
In Wales, places of worship and retail stores reopened, and restaurants resumed outdoor service. Outdoor wedding receptions with up to 30 people can take place.
Cases remain low in Britain, with more than 40 percent of the population having received at least one dose of a vaccine. On Sunday, the country reported just over 1,700 new cases and 11 deaths, according to a New York Times database.
 Credit…Stephen Zenner/Getty Images
Credit…Stephen Zenner/Getty Images
West Virginia will give $100 savings bonds to 16- to 35-year-olds who get a Covid-19 vaccine, Gov. Jim Justice said on Monday.
There are roughly 380,000 West Virginians in that age group, many of whom have already gotten at least one shot, but Mr. Justice said he hoped the money would motivate the rest to get inoculated, as “they’re not taking the vaccines as fast as we’d like them to take them.”
The state will use federal funds from the CARES Act to pay for the bonds, Mr. Justice, a Republican, said at a news conference, adding that he had “vetted this every way that we possibly can” to ensure that the unconventional use of the funds was allowed.
The bonds will be also be available to anyone in that age group who has already been vaccinated, Mr. Justice said.
West Virginia has the 16th highest rate of new coronavirus cases per person among U.S. states and ranks 12th in hospitalizations, according to a New York Times database.
Mr. Justice said the state needed to stop the virus “dead in its tracks,” and that if it did, “these masks go away, the hospitalizations go away, the death toll and the body bags start to absolutely become minimal.”
Earlier this year, at the start of the country’s vaccination effort, West Virginia had stood out for its success in vaccinating its residents. At one point, it had administered second doses to more of its population than any other state; it was also behind only Alaska for the percent of its residents that had received a first dose.
But now West Virginia is fallen behind, ahead of only nine states for the portion of its residents that have had a first dose, according to a New York Times database tracking vaccines.
Mr. Justice said that young West Virginians could “always stand an extra dose of patriotism.” He urged them to “accept that wonderful savings bond” — which will allow the recipient to retrieve the $100, plus interest, at a later date — adding, “I hope that you keep it for a long, long, long time.”
 Credit…Pool photo by Becky Bohrer
Credit…Pool photo by Becky Bohrer
Alaska Airlines has suspended an Alaska state lawmaker from its flights for violating its mask policies, the company said.
Lora Reinbold, a Republican state senator, was arguing with employees at Juneau International Airport about the airline’s mask rules, according to footage posted on Twitter.
“We need you to pull the mask up, or I’m not going to let you on the flight,” an employee is heard saying to Ms. Reinbold on the videos, which were posted on Thursday.
“It is up,” Ms. Reinbold responds.
“It is not,” an employee says. “It’s down below your nose. We can’t have it down.”
The airline said it had told Ms. Reinbold that she was “not permitted to fly with us for her continued refusal to comply with employee instruction regarding the current mask policy,” adding that the suspension is being reviewed.
The clash over the company’s rule was the latest to surface in the country about masks during the pandemic. Mask mandates have become a rallying cry for some activists and a divisive political talking point. Disputes about the rules have sometimes led to angry confrontations.
 Credit…Charlie Riedel/Associated Press
Credit…Charlie Riedel/Associated Press
U.S. airlines have been bolstered by the return of customers eager to travel within the country or just outside its borders, but the nation’s largest carriers are still lamenting the loss of two particularly lucrative parts of the business: international and corporate travel. At least one of those could rebound this summer.
In an interview with The New York Times over the weekend, Ursula von der Leyen, the president of the European Commission, said she expected the European Union to ease travel restrictions for vaccinated American tourists, a move that could let the airline industry cash in during the year’s busiest travel season.
“Long-haul international flying represents a significant opportunity for United,” Andrew Nocella, the chief commercial officer for United Airlines, told investors last week. “We have seen in recent weeks that immediately after a country provides access with proof of a vaccine, leisure demand returns to the level of 2019 quickly.”
American Airlines and United said this month that international travel remained about 80 percent lower than in 2019. They and other airlines expect strong demand for domestic flights this summer, and the restoration of trans-Atlantic travel could provide the industry a much-needed boost as it works to generate profits again.
American, Delta Air Lines and United each reported a loss of more than $1 billion in the first three months of the year. Southwest Airlines reported a small profit, of $116 million, though its chief executive said the airline would have lost $1 billion without federal aid.
The news of the E.U. reopening to vaccinated American tourists was also welcomed by Willie Walsh, the director general of the International Air Transport Association, a global airline industry group, who said it could bode well for carriers elsewhere, too.
He said in a statement that coordination between the European Commission and the industry was essential “so that airlines can plan within the public health benchmarks and timelines that will enable unconditional travel for those vaccinated,” not just Americans but passengers from other countries as well.
 Credit…Adam Dean for The New York Times
Credit…Adam Dean for The New York Times
Only a few weeks ago, Phuket seemed poised for a comeback. After a year of practically no foreign tourists arriving in Thailand, the national government decided that Phuket would start welcoming vaccinated visitors in July, without requiring them to go through quarantine. The project was called Phuket Sandbox.
But Thailand is now gripped by its worst Covid-19 outbreak since the pandemic began, spread in part by the well-heeled Thais who partied in Phuket and Bangkok with no social distancing. The confirmed daily caseload — albeit low by global standards — has increased from 26 on April 1 to more than 2,000 three weeks later, in a country that in early December had about 4,000 cases total.
The opening that Phuket had planned for July 1 now appears unlikely, Thailand’s tourism minister acknowledged this month.
“If you ask me how optimistic I am, I cannot say,” said Nanthasiri Ronnasiri, the director of the tourism authority’s Phuket office. “The situation changes all the time.”
The virus’s resurgence after so many months of economic hardship is devastating for the majority of Phuket’s residents, who depend on foreign tourists for their livelihoods.
 Credit…Lynne Sladky/Associated Press
Credit…Lynne Sladky/Associated Press
A private school in the fashionable Design District of Miami sent its faculty and staff a letter last week about getting vaccinated against Covid-19. But unlike institutions that have encouraged and even facilitated vaccination for teachers, the school, Centner Academy, did the opposite: One of its co-founders, Leila Centner, informed employees “with a very heavy heart” that if they chose to get a shot, they would have to stay away from students.
In an example of how misinformation threatens the nation’s effort to vaccinate enough Americans to get the coronavirus under control, Ms. Centner, who has frequently shared anti-vaccine posts on Facebook, claimed in the letter that “reports have surfaced recently of non-vaccinated people being negatively impacted by interacting with people who have been vaccinated.”
“Even among our own population, we have at least three women with menstrual cycles impacted after having spent time with a vaccinated person,” she wrote, repeating a false claim that vaccinated people can somehow pass the vaccine to others and thereby affect their reproductive systems. (They can do neither.)
In the letter, Ms. Centner gave employees three options:
-
Inform the school if they had already been vaccinated, so they could be kept physically distanced from students;
-
Let the school know if they get the vaccine before the end of the school year, “as we cannot allow recently vaccinated people to be near our students until more information is known”;
-
Wait until the school year is over to get vaccinated.
Teachers who get the vaccine over the summer will not be allowed to return, the letter said, until clinical trials on the vaccine are completed, and then only “if a position is still available at that time” — effectively making teachers’ employment contingent on avoiding the vaccine.
Credit…Romain Maurice/Getty Images for Haute Living
Ms. Centner required the faculty and staff to fill out a “confidential” form revealing whether they had received a vaccine — and if so, which one and how many doses — or planned to get vaccinated. The form requires employees to “acknowledge the School will take legal measures needed to protect the students if it is determined that I have not answered these questions accurately.”
Ms. Centner directed questions about the matter to her publicist, who said in a statement that the school’s top priority throughout the pandemic has been to keep students safe. The statement repeated false claims that vaccinated people “may be transmitting something from their bodies” leading to adverse reproductive issues among women.
“We are not 100 percent sure the Covid injections are safe and there are too many unknown variables for us to feel comfortable at this current time,” the statement said.
The Food and Drug Administration, the Centers for Disease Control and Prevention, the World Health Organization and many other authorities have concluded that the coronavirus vaccines now in emergency use in the United States are safe and effective.
The Centner Academy opened in 2019 for students in prekindergarten through eighth grade, promoting itself as a “happiness school” focused on children’s mindfulness and emotional intelligence. The school prominently advertises on its website support for “medical freedom from mandated vaccines.”
Ms. Centner founded the school with her husband, David Centner, a technology and electronic highway tolling entrepreneur. Each has donated heavily to the Republican Party and the Trump re-election campaign, while giving much smaller sums to local Democrats.
In February, the Centners welcomed a special guest to speak to students: Robert F. Kennedy Jr., the prominent antivaccine activist. (Mr. Kennedy was suspended from Instagram a few days later for promoting Covid-19 vaccine misinformation.) This month, the school hosted a Zoom talk with Dr. Lawrence Palevsky, a New York pediatrician frequently cited by anti-vaccination activists.
Kitty Bennett contributed research.
 Credit…Gianni Cipriano for The New York Times
Credit…Gianni Cipriano for The New York Times
The number of students that dropped out of school in Italy because of the coronavirus pandemic is rising, aggravating what was already a crisis before the disease spread across the nation.
Italy had among the worst dropout rates in the European Union, and the southern city of Naples was particularly troubled by high numbers. When the coronavirus hit, Italy shuttered its schools more than just about all the other European Union member states, with especially long closures in the Naples region, pushing students out in even higher numbers.
While it is too early for reliable statistics, principals, advocates and social workers say they have seen a sharp increase in the number of students falling out of the system. The impact on an entire generation may be one of the pandemic’s lasting tolls.
Italy closed its schools — fully or in part — for 35 weeks in the first year of the pandemic — three times longer than France, and more than Spain or Germany.
And experts say that by doing so, the country, which has Europe’s oldest population and was already lagging behind in critical educational indicators, has risked leaving behind its youth, its greatest and rarest resource for a strong post-pandemic recovery.