Here’s what you need to know:
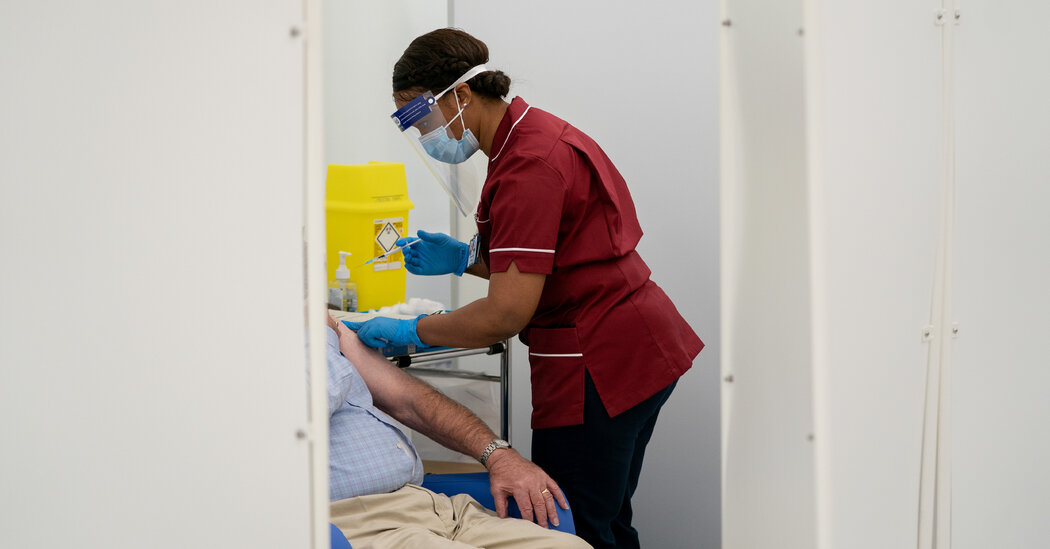
Goods are piling up at some British ports, as trucks and rail have failed to keep up with companies trying to stockpile ahead of Brexit.Credit…Andrew Testa for The New York Times
Businesses in Britain and the European Union are bracing for the economic disruption of Brexit, which threatens to clog ports and disrupt trade across the English Channel on Dec. 31 if leaders do not reach a compromise to settle their future trading relationship.
But the economic breakup could have a relatively limited impact on trade with the United States, trade experts said.
Because the United States does not have a free-trade agreement with the European Union, Britain’s departure from the bloc will do little to alter its trading relationship with the United States. Following Brexit, the terms of trade between the United States and Britain will continue to be governed by the rules of the World Trade Organization, as they were before.
The direct effect on the two trade partners “should be minimal given there’s no change in tariffs,” said Christopher Rogers, a global trade and logistics analyst at Panjiva.
Still, he said, significant customs disruptions between Europe and Britain could have knock-on effects for supply chains, if, for example, it takes British businesses that are exporting to the United States longer to source components from abroad. Goods are piling up at some British ports, as trucks and rail have failed to keep up with companies trying to stockpile ahead of Brexit.
Britain’s trading terms with the United States may not get much worse, but they also appear unlikely to get better.
The two countries have been carrying out negotiations for a free-trade deal since May. But with the election of Joseph R. Biden Jr., the prospects for that agreement, which many Britons saw as a source of post-Brexit strength, have been greatly diminished.
The congressional authority that gives trade deals an easier path to approval by Congress, called trade promotion authority, is set to expire this summer, and Mr. Biden has promised not to enter into any major new trade agreements until the United States has made major investments at home.
 Boeing 737 Max aircraft in a lot at Boeing Field in Seattle. The plane was grounded worldwide almost two years ago.Credit…Lindsey Wasson/Reuters
Boeing 737 Max aircraft in a lot at Boeing Field in Seattle. The plane was grounded worldwide almost two years ago.Credit…Lindsey Wasson/Reuters
Gol Airlines, a Brazilian carrier, said it planned to start flights aboard the Boeing 737 Max on Wednesday, making it the first airline to fly passengers on the plane since it was grounded worldwide almost two years ago.
The first flights will be on domestic routes to and from Gol’s hub in São Paulo, with the company expecting all seven of the Max planes in its fleet to be updated and cleared to fly by the end of the month. A Gol spokeswoman declined to provide further details.
“Our first priority is always the safety of our customers,” Celso Ferrer, vice president of operations and a commercial pilot at Gol, said in a statement. “Over the past 20 months, we have watched the most comprehensive safety review in the history of commercial aviation unfold.”
The Max was banned worldwide in March 2019 after a total of 346 people were killed in two crashes aboard the plane. In the United States, the Federal Aviation Administration last month became the first regulator to allow the plane to fly again, after required modifications are made. The agency was recently joined by regulators in Brazil, while the European aviation authority has suggested that it plans to lift its ban within weeks. Relatives of those killed in the crashes criticized the decision to allow the plane to fly again, arguing that it remains unsafe.
The lifting of the ban allows Boeing to restart sales and deliveries in earnest after its passenger airline business was pummeled by the grounding and the pandemic. The plane maker on Tuesday reported a net decline of 61 orders last month. Boeing’s backlog of orders, most of them for the Max, stood at 4,240, down more than a thousand from the start of the year after accounting for fulfilled orders.
Still, airlines are still interested in acquiring the plane. Last week, the company announced it had agreed to sell 75 Max jets to Ryanair, the low-cost European airline. Like RyanAir, Gol is among the biggest customers for the Max. The airline’s fleet is composed of 127 Boeing planes and it has an order for 95 Max jets scheduled for delivery over a decade starting in 2022.
 Brian Chesky, Airbnb’s co-founder, in 2018. It’s usually not regular people, employees or even pre-I.P.O. investors who get a windfall from initial public offerings.Credit…Eric Risberg/Associated Press
Brian Chesky, Airbnb’s co-founder, in 2018. It’s usually not regular people, employees or even pre-I.P.O. investors who get a windfall from initial public offerings.Credit…Eric Risberg/Associated Press
A dirty secret of initial public offerings is that even the coolest ones may make only a handful of people rich — and it may not be regular people, employees or even fancy pre-I.P.O. investors who get a windfall.
DoorDash and Airbnb are expected to have spectacular first sales on public stock exchanges this week and start trading at far higher levels than anticipated even a few weeks ago.
But buying stock in relatively young and unproven companies — which usually describes technology companies selling their stock to the public for the first time — is often a coin-toss bet. Even the professional investors who buy stock in hot companies before they go public don’t always get rich, unless they throw their money around early and get lucky. Companies you might have heard of like Uber, Lyft, Snapchat and Slack were at best meh I.P.O. investments.
Look at Airbnb. Among the investors who got a special chance to buy Airbnb stock nearly four years ago, each $10,000 of stock they bought will be worth about $11,500 if Airbnb starts selling its shares to the public for $60 each. Nice!
But if your aunt had invested $10,000 nearly four years ago in a simple fund that mirrored the ups and downs of the S&P 500 stock index, she would now have $15,600. Even nicer.
The pandemic hurt business for Uber and Lyft, but their stocks were losers before then. Uber’s stock price has bounced back and is now up 30 percent since the spring, and still anyone who bought Uber shares in its 2019 I.P.O. — and even the professional investors who bought its stock in the four years before that — would have made far more money buying an index fund. Uber employees who were hired before the I.P.O. and were paid partly in stock also would have been better off getting paid in an index fund.
People who bought Snapchat’s stock in its 2017 initial public offering had to wait more than three years to not lose money on their bet. Slack just sold itself at a share price not much higher than its first public stock sale last year.
These are cherry-picked examples. There are companies whose stock prices have soared since their I.P.O.s and made people rich — Zoom Video is a prominent example in technology. And the people who have already bet on the restaurant delivery app DoorDash stand to make a big profit when the company goes public this week.
Will Airbnb be a winning I.P.O.? It depends. It definitely will be for the venture capital firm Sequoia, which bet on Airbnb early. And it’s certainly faring better than people expected when travel froze early this year. But no one can confidently predict whether its share price will shoot to the moon like Zoom’s has since its 2019 I.P.O. or will plunge as Lyft’s did after it went public.
That’s the lesson. Cool companies don’t always make good investments. The people screaming on Robinhood about their splurge on a hot I.P.O. may not know what they’re talking about.
By: Ella Koeze·Source: Refinitiv
-
Stocks were unsteady on Tuesday, as the spread of coronavirus cases and restrictions on people’s movement and businesses outweighed optimism about the rollout of a vaccine.
-
The S&P 500 was flat by midday after recovering from an earlier dip. The Stoxx Europe 600 and Britain’s FTSE 100 also recouped small losses and were slightly higher.
-
In the United States, rising numbers of virus cases has led California to impose new stay-at-home orders in large swathes of the state. In New York, the number of people hospitalized with the coronavirus is rising and could lead to another ban on indoor dining.
-
In Europe, countries are struggling to emerge from a second wave of the pandemic. The infection rate in France is threatening plans to ease restrictions before the holidays, and in Greece, the lockdown was extended until early January.
-
But on a brighter note, Britain on Tuesday started a mass vaccination campaign, delivering the first shots of the Pfizer-BioNTech Covid-19 vaccine. “There is finally some clear light at the end of a very dark tunnel,” James Pomeroy, an economist at HSBC, wrote in a note to clients. “And that cheer should be seen in some of the economic data in the coming year too.”
-
Tesla said on Tuesday it would sell as much as $5 billion in shares, its third return to markets in 10 months, and use the money for more investments including factory construction. Tesla’s shares were down nearly 3 percent. This year, the electric carmaker’s shares have risen about 670 percent, and later this month, the company will join the S&P 500.
 Google’s offices in London. Britain’s top antitrust regulator recommended a new tech watchdog.Credit…Ben Stansall/Agence France-Presse — Getty Images
Google’s offices in London. Britain’s top antitrust regulator recommended a new tech watchdog.Credit…Ben Stansall/Agence France-Presse — Getty Images
Governments around the world have been grappling with ways to crimp the power of the biggest tech companies. In the United States, the Justice Department recently filed an antitrust case against Google. The European Union has issued antitrust violations and enacted stiffer data-protection laws. The Australian government is pushing new rules to make Google and Facebook pay for certain content.
But many question whether the tactics are adequate, particularly if a lengthy enforcement and legal process slows down action against the fast-moving and deep-pocketed companies.
On Tuesday, Britain’s top antitrust regulator recommended a new approach. The Competition and Markets Authority released recommendations for creating a new regulator called the Digital Markets Unit that will focus on the biggest technology platforms. The regulator would be able to fine companies up to 10 percent of global revenue.
The idea of creating a tech industry regulator has gained momentum among academics and policymakers around the world. The aim is to treat giants like Amazon, Apple, Facebook, Google, and Microsoft more like the biggest companies in banking and health care — with dedicated regulators that have the expertise in the subject matter to serve as a watchdog and act quickly to address wrongdoing, akin to the Securities and Exchange Commission and the Food and Drug Administration.
Britain is perhaps the furthest along. The new regulator would be responsible for enforcing a legally binding code of conduct intended to prevent the biggest companies from using their dominance to exploit consumers and business, or to box out emerging competitors. Officials said only companies of a certain size would fall under the rules, which would be tailored to specific types of businesses. Google and Facebook may face certain restrictions related to digital advertising, while Amazon would have others related to e-commerce.
To improve competition, the regulator could force companies to share certain data with rivals, and it would review acquisitions.
The proposals build on recommendations made by a British panel of experts last year and are part of a process by the government to enact regulations for the digital economy by next year. Britain is preparing to leave the European Union, which next week will release its own draft laws to increase oversight of the tech industry across the 27-nation bloc.
British authorities have raised specific concerns about the digital advertising market dominated by Google and Facebook. In July, the Competition and Markets Authority published a 437-page investigation that concluded the two companies have such scale and unmatched access to user data that “potential rivals can no longer compete on equal terms.”
Goldman Sachs has reached a deal to buy out the minority partner in its Chinese securities joint venture, which could make it the first global bank to assume full ownership of its securities business in mainland China since the Communist Party took control of foreign-owned enterprises in the country in the 1950s.
In a memo to employees on Tuesday, the Wall Street bank said it had reached a definitive agreement to buy a 49 percent stake in Goldman Sachs Gao Hua still held by its local partner, Beijing Gao Hua Securities. Goldman Sachs did not disclose a price for the transaction.
The deal follows a pledge by Chinese leaders in 2017, amid worsening trade relations with the United States, to relax or remove limits on foreign bank ownership. The move was part of an unsuccessful effort by China to enlist Wall Street in heading off President Trump’s plans to impose tariffs on Chinese goods.
Goldman Sachs could be the first to take full control of its China securities business, depending on regulatory approval and how quickly the deal is completed.
JP Morgan Chase already has full ownership of its futures business in China, but still has a joint venture for other activities on the mainland. Other investment banks, like JP Morgan Chase, Morgan Stanley, UBS and Nomura, are in various stages of raising their stakes in their Chinese securities operations.
Commercial banks, by contrast, have avoided raising their stakes in commercial banking operations in mainland China above 25 percent. Doing so would subject those operations to further global banking regulations.
Goldman Sachs had announced on March 27 that it had obtained regulatory approval to raise its stake in Goldman Sachs Gao Hua from 33 percent to 51 percent. Tuesday’s memo was reported earlier by The Wall Street Journal.
 With movie theaters largely shut across the United States, traditional movie companies like Warner Bros. are being forced to evolve.Credit…Aaron P/Bauer-Griffin, via Getty
With movie theaters largely shut across the United States, traditional movie companies like Warner Bros. are being forced to evolve.Credit…Aaron P/Bauer-Griffin, via Getty
Last week, when Jason Kilar, WarnerMedia’s chief executive, announced that 17 more Warner Bros. movies would each roll out on HBO Max and in theaters simultaneously. To prevent the news of the 17-movie shift from leaking (and to make the move speedily rather than get mired in the expected blowback), WarnerMedia kept the major agencies and talent management companies in the dark until roughly 90 minutes before issuing a news release, report Brooks Barnes and Nicole Sperling.
The surprise move left agencies on a war footing. Representatives for major Warner Bros. Talk of a Warner Bros. boycott began circulating inside the Directors Guild of America. A partner at one talent agency spent part of the weekend meeting with litigators. Some people started to angrily refer to the studio as Former Bros.
The 97-year-old studio, the ancestral home of Humphrey Bogart (“Casablanca”) and Bette Davis (“Now, Voyager”), suddenly finds itself at the uncomfortable center of a Hollywood that is changing at light speed. Even before the pandemic, streaming services like Netflix, Apple TV+ and Amazon Prime Video were upending how movies get seen and their creators are compensated. Now, with theaters struggling because of the coronavirus and the public largely stuck at home, even traditional film companies are being forced to evolve.
It’s not that all actors and directors are against streaming. Plenty of big names are making movies for Netflix. But last week’s move by Warner Bros. raised fundamental financial questions. If old-line studios are no longer trying to maximize the box office for each film but instead shifting to a hybrid model where success is judged partly by ticket sales and partly by the number of streaming subscriptions sold, what does that mean for talent pay packages?
How studios compensate A-list actors, directors, writers and producers is complicated, with contracts negotiated film by film and person by person. But it boils down to two checks. One is guaranteed (a large upfront fee) and one is a gamble: a portion of ticket sales after the studio has recouped its costs.
If a film flops, the second payday never comes. If a film is a hit, as is often the case with superheroes and other fantasy stories, the “back end” pay can add up to wheelbarrows full of cash.
 A garage at the Aurora office in Palo Alto, Calif.Credit…Jason Henry for The New York Times
A garage at the Aurora office in Palo Alto, Calif.Credit…Jason Henry for The New York Times
Uber, which spent hundreds of millions of dollars on a self-driving car project that executives once believed was a key to becoming profitable, is handing the autonomous vehicle effort over to a Silicon Valley start-up, the companies said on Monday.
Uber will also invest $400 million in the start-up, called Aurora, so it is essentially paying the company to take over the autonomous car operation, which had become a financial and legal headache. Uber is likely to license whatever technology Aurora manages to create.
The deal amounts to a fire-sale end to a high-profile but star-crossed effort to replace Uber’s human drivers with machines that could drive on their own. It is also indicative of the challenges facing other autonomous vehicle projects, which have received billions in investments from Silicon Valley and automakers but have not produced the fleets of robotic vehicles some thought would be on the streets by now.
Aurora’s chief executive, Chris Urmson, said Aurora’s first product will not be a robot taxi that could help with Uber’s ride-hailing business. Instead, it will likely be a self-driving truck, which Mr. Urmson believes has a better chance of success in the near term because long-haul truck driving on highways is more predictable and does not involve passengers.
In a statement, the Uber chief executive, Dara Khosrowshahi, said he was looking forward to bringing Aurora technology to market “in the years ahead.” Uber declined to comment further on the agreement.
-
Rashida Jones, a senior vice president for news at MSNBC and NBC News, will become the first Black woman to take charge of a major television news network. Her promotion, announced by Cesar Conde, the chairman of NBCUniversal News Group, is another big shake-up in the network’s management ranks. She will succeed Phil Griffin, the MSNBC president whose left-leaning shows yielded big ratings in the Trump years and minted media brands like “The Rachel Maddow Show” and “Morning Joe,” will depart on Feb. 1 after a 12-year tenure, the network said on Monday.
-
The Japanese advertising giant Dentsu Group plans to cut roughly 6,000 jobs as it grapples with the effects of the coronavirus pandemic. In a securities filing in Tokyo on Monday, Dentsu laid out details of its restructuring strategy, which will cost 88 billion yen (about $850 million) to carry out over two years and includes trimming its 48,000-person international work force by 12.5 percent. The timeline will vary by location, the company said.
Patrick Gaspard, a former aide to President Barack Obama, U.S. ambassador to South Africa and executive director of the Democratic National Committee, has emerged as the leading candidate to be nominated as labor secretary under President-elect Joseph R. Biden Jr., according to people with knowledge of the discussions.
Mr. Gaspard announced last week that he would step down as the head of the Open Society Foundations, founded by the liberal megadonor George Soros, at the end of the year, fueling speculation in Washington that he was poised to join the incoming administration. He has a background in labor organizing, including a senior leadership position for the Service Employees International Union, which he held before joining the Obama administration.
His potential nomination would give Mr. Biden, who calls himself a “union guy,” a labor secretary with union roots. He would also add to the list of Black cabinet appointees, a key goal of Mr. Biden’s transition team as it seeks to fulfill Mr. Biden’s campaign promise of diversity in the top leadership of his administration.
Born in the Democratic Republic of Congo to Haitian parents, Mr. Gaspard immigrated to the United States in early childhood, grew up in New York and attended Columbia University before leaving to work on Jesse Jackson’s 1988 presidential campaign. He worked for years in New York City politics and on Howard Dean’s 2004 Democratic presidential bid, and he was an aide to former Mayor David Dinkins. After Mr. Dinkins died last month, Mr. Gaspard wrote on Twitter, “He taught me that you don’t need to be loud to be strong.”
Mr. Gaspard worked for years as an organizer and rose through the Service Employees International Union to become its national political director before joining Mr. Obama’s 2008 presidential campaign. In the Obama White House, Mr. Gaspard served as director of political affairs, before helming the Democratic National Committee and being confirmed as Mr. Obama’s ambassador to South Africa.
Allies of Senator Bernie Sanders, independent of Vermont and Mr. Biden’s chief rival for the Democratic nomination this year, had pushed hard for Mr. Sanders to be selected as labor secretary. But Mr. Biden’s short list for the job does not appear to include Mr. Sanders.