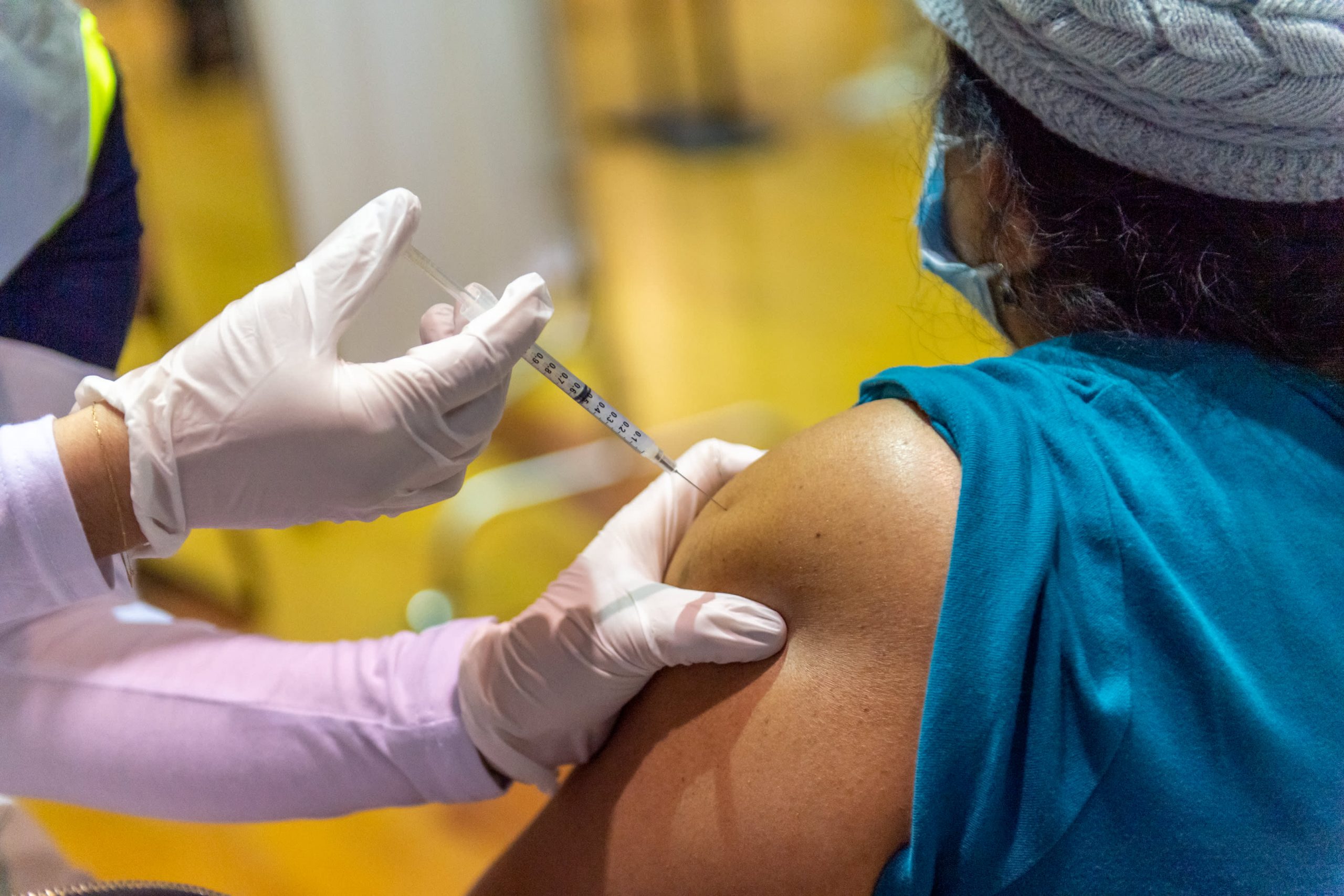
More than 20,000 people were vaccinated against Covid-19 last weekend at Bank of America Stadium in Charlotte, North Carolina. The idea for the three-day event came during a humble walk, according to Darius Adamczyk, CEO of Honeywell International.
“In the Covid era, one of the more social things you can still do is go for a walk outside with some of your friends,” Adamczyk said on Squawk Box on Tuesday. One weekend, Adamczyk said he was walking with Carolina Panthers President Tom Glick and Atrium Health CEO Gene Woods, who both live in his neighborhood.
The men discussed the introduction of Covid vaccinations in the US, which started more slowly than expected from mid-December, Adamczyk recalled. “We said, ‘You know, maybe we could help here. Maybe we could work together as a team.'”
Atrium Health, as a non-profit healthcare system with 42 hospitals, could of course direct the actual administration of the vaccines, Adamczyk said. The Panthers are now well experienced in handling large crowds at Bank of America Stadium, where David Tepper’s NFL franchise plays its home games.
Honeywell could bring its logistics and sales expertise, as well as its technological capabilities, to the table more broadly, Adamczyk said. Put all three Charlotte-based organizations together, he said, and “we think we can do something really different.”
“I have to thank our mayor, [Vi Alexander Lyles,] thank our governor, [Roy Cooper,] for actually shooting ourselves because it could have been a disaster, “said Adamczyk. But it turned out to be a success, he said.
The goal was to deliver 19,000 vaccines at the stadium event, a spokesman for Atrium Health told CNBC. In the end, more than 20,000 were administered. The week before, Honeywell, Atrium Health, and Tepper Sports & Entertainment, the company that holds Tepper’s ownership of the Panthers, also worked together on a vaccination site at Charlotte Motor Speedway, where more than 15,000 shots were fired.
The pace of vaccinations in the US has improved in recent weeks and the number of doses given now exceeds the number of confirmed Covid cases since the pandemic began. As of Monday, a total of 32.8 million doses had been administered, according to the Centers for Disease Control and Prevention, including just over 6 million Americans who both received two-dose vaccinations. 26.4 million coronavirus cases have been confirmed in the United States, data from Johns Hopkins University shows.
The event at Bank of America Stadium was vaccinated every 4.5 seconds on average, Adamczyk said. “The other statistic that I think is really important here is that 30% are from communities of colored people.”
“We did it in three days – Friday, Saturday, Sunday,” he added. “Twelve hours a day, 20,000 people. See if we could do it and set up 50 or 100 such locations across the country.”
Adamczyk acknowledged that vaccine supply restrictions may currently prohibit this vision, but was confident that those restrictions would ease in the coming weeks and months.
“Ultimately, this becomes a queuing problem, and the right and most efficient way to solve the queuing problem is to have very large, very efficient distribution centers that are all over the country, across the states, and very quickly take them in the arms of the people, “said Adamczyk.
“We have to get back to life, we have to go back to good economic times and the fastest way the economy can recover is to get people vaccinated,” he added.