Durch die Monopolisierung der Impfstoffversorgung gegen Covid-19 drohen den reichen Nationen mehr als eine humanitäre Katastrophe: Die daraus resultierenden wirtschaftlichen Verwüstungen werden die wohlhabenden Länder fast genauso hart treffen wie die in den Entwicklungsländern.
Dies ist die entscheidende Erkenntnis aus einer akademischen Studie, die am Montag veröffentlicht werden soll. Im extremsten Szenario – mit reichen Nationen, die bis Mitte dieses Jahres vollständig geimpft waren, und armen Ländern, die weitgehend geschlossen sind – kommt die Studie zu dem Schluss, dass die Weltwirtschaft Verluste von mehr als 9 Billionen US-Dollar erleiden würde, eine Summe, die über der Jahresproduktion Japans und Deutschlands liegt kombiniert.
Fast die Hälfte dieser Kosten würde von wohlhabenden Ländern wie den Vereinigten Staaten, Kanada und Großbritannien übernommen.
In dem Szenario, das Forscher als am wahrscheinlichsten bezeichnen und in dem Entwicklungsländer bis Ende des Jahres die Hälfte ihrer Bevölkerung impfen, würde die Weltwirtschaft immer noch einen Schlag zwischen 1,8 und 3,8 Billionen US-Dollar absorbieren. Mehr als die Hälfte der Schmerzen würde sich auf wohlhabende Länder konzentrieren.
Die von der Internationalen Handelskammer in Auftrag gegebene Studie kommt zu dem Schluss, dass eine gerechte Verteilung der Impfstoffe im wirtschaftlichen Interesse jedes Landes liegt, insbesondere derjenigen, die am stärksten vom Handel abhängen. Es ist eine Zurechtweisung für die weit verbreitete Auffassung, dass der Austausch von Impfstoffen mit armen Ländern lediglich eine Form der Wohltätigkeit ist.
“Natürlich sind alle Volkswirtschaften miteinander verbunden”, sagte Selva Demiralp, Wirtschaftswissenschaftlerin an der Koc-Universität in Istanbul, die zuvor bei der Federal Reserve in Washington gearbeitet hatte, und eine der Autoren der Studie. “Keine Volkswirtschaft wird vollständig wiederhergestellt, wenn die anderen Volkswirtschaften nicht wiederhergestellt werden.”
Frau Demiralp merkte an, dass eine globale philanthropische Initiative namens ACT Accelerator, die darauf abzielt, Entwicklungsländern Pandemieressourcen zur Verfügung zu stellen, Verpflichtungen in Höhe von weniger als 11 Milliarden US-Dollar für ein Ziel von 38 Milliarden US-Dollar eingegangen ist. Die Studie legt die wirtschaftlichen Gründe für das Schließen der Lücke dar. Die verbleibenden 27 Milliarden US-Dollar mögen auf den ersten Blick wie eine enorme Summe aussehen, sind aber im Vergleich zu den Kosten für die Fortsetzung der Pandemie eine Kleinigkeit.
Die alltägliche Idee, dass die Pandemie weder Grenzen noch Rassen- und Klassenunterschiede respektiert, wurde von Unternehmensleitern und Experten gefördert. Dieses tröstliche Konzept wurde durch die Tatsache widerlegt, dass Covid-19 den Tod und die Zerstörung von Lebensgrundlagen bei Niedriglohn-Servicemitarbeitern und insbesondere bei rassistischen Minderheiten trainiert hat, während Angestellte weitgehend sicher von zu Hause aus arbeiten konnten, und einige andere der reichsten Menschen der Welt können die Pandemie auf Yachten und Privatinseln ausreiten.
Aber im Bereich des internationalen Handels gibt es kein Versteck vor dem Coronavirus, wie die Studie zeigt. Stattdessen gibt es globale Lieferketten, die die Teile für die Industrie produzieren und die weiterhin gestört werden, solange das Virus eine Kraft bleibt.
Ein Team von Wirtschaftswissenschaftlern der Koc University, der Harvard University und der University of Maryland untersuchte Handelsdaten in 35 Branchen in 65 Ländern und untersuchte ausführlich die wirtschaftlichen Auswirkungen einer ungleichen Impfstoffverteilung.
Covid19 Impfungen >
Antworten auf Ihre Impfstofffragen
Wenn ich in den USA lebe, wann kann ich den Impfstoff bekommen?
Während die genaue Reihenfolge der Impfstoffempfänger von Staat zu Staat unterschiedlich sein kann, werden die meisten Ärzte und Bewohner von Langzeitpflegeeinrichtungen an erster Stelle stehen. Wenn Sie verstehen möchten, wie diese Entscheidung getroffen wird, hilft dieser Artikel.
Wann kann ich nach der Impfung wieder zum normalen Leben zurückkehren?
Das Leben wird erst wieder normal, wenn die Gesellschaft als Ganzes ausreichend Schutz gegen das Coronavirus erhält. Sobald die Länder einen Impfstoff zugelassen haben, können sie in den ersten Monaten höchstens einige Prozent ihrer Bürger impfen. Die nicht geimpfte Mehrheit bleibt weiterhin anfällig für Infektionen. Eine wachsende Anzahl von Coronavirus-Impfstoffen zeigt einen robusten Schutz vor Krankheit. Es ist aber auch möglich, dass Menschen das Virus verbreiten, ohne zu wissen, dass sie infiziert sind, weil sie nur leichte oder gar keine Symptome haben. Wissenschaftler wissen noch nicht, ob die Impfstoffe auch die Übertragung des Coronavirus blockieren. Selbst geimpfte Menschen müssen vorerst Masken tragen, Menschenmassen in Innenräumen meiden und so weiter. Sobald genügend Menschen geimpft sind, wird es für das Coronavirus sehr schwierig, gefährdete Personen zu finden, die infiziert werden können. Je nachdem, wie schnell wir als Gesellschaft dieses Ziel erreichen, könnte sich das Leben im Herbst 2021 einem normalen Zustand nähern.
Muss ich nach der Impfung noch eine Maske tragen?
Ja, aber nicht für immer. Die beiden Impfstoffe, die möglicherweise in diesem Monat zugelassen werden, schützen die Menschen eindeutig vor einer Krankheit mit Covid-19. Die klinischen Studien, die diese Ergebnisse lieferten, waren jedoch nicht darauf ausgelegt, festzustellen, ob geimpfte Personen das Coronavirus noch verbreiten können, ohne Symptome zu entwickeln. Das bleibt eine Möglichkeit. Wir wissen, dass Menschen, die von Natur aus mit dem Coronavirus infiziert sind, es verbreiten können, ohne Husten oder andere Symptome zu haben. Die Forscher werden diese Frage bei der Einführung der Impfstoffe intensiv untersuchen. In der Zwischenzeit müssen sich selbst geimpfte Menschen als mögliche Spreizer vorstellen.
Wird es wehtun? Was sind die Nebenwirkungen?
Der Impfstoff gegen Pfizer und BioNTech wird wie andere typische Impfstoffe als Schuss in den Arm abgegeben. Die Injektion unterscheidet sich nicht von denen, die Sie zuvor erhalten haben. Zehntausende Menschen haben die Impfstoffe bereits erhalten, und keiner von ihnen hat ernsthafte gesundheitliche Probleme gemeldet. Einige von ihnen haben jedoch kurzlebige Beschwerden verspürt, darunter Schmerzen und grippeähnliche Symptome, die normalerweise einen Tag anhalten. Es ist möglich, dass die Leute planen müssen, nach dem zweiten Schuss einen Tag frei zu nehmen oder zur Schule zu gehen. Obwohl diese Erfahrungen nicht angenehm sind, sind sie ein gutes Zeichen: Sie sind das Ergebnis der Begegnung Ihres eigenen Immunsystems mit dem Impfstoff und einer starken Reaktion, die eine dauerhafte Immunität gewährleistet.
Werden mRNA-Impfstoffe meine Gene verändern?
Nein. Die Impfstoffe von Moderna und Pfizer verwenden ein genetisches Molekül, um das Immunsystem zu stärken. Dieses als mRNA bekannte Molekül wird schließlich vom Körper zerstört. Die mRNA ist in einer öligen Blase verpackt, die mit einer Zelle verschmelzen kann, so dass das Molekül hineinrutschen kann. Die Zelle verwendet die mRNA, um Proteine aus dem Coronavirus herzustellen, die das Immunsystem stimulieren können. Zu jedem Zeitpunkt kann jede unserer Zellen Hunderttausende von mRNA-Molekülen enthalten, die sie produzieren, um eigene Proteine herzustellen. Sobald diese Proteine hergestellt sind, zerkleinern unsere Zellen die mRNA mit speziellen Enzymen. Die mRNA-Moleküle, die unsere Zellen herstellen, können nur wenige Minuten überleben. Die mRNA in Impfstoffen ist so konstruiert, dass sie den Enzymen der Zelle etwas länger standhält, sodass die Zellen zusätzliche Virusproteine bilden und eine stärkere Immunantwort auslösen können. Die mRNA kann jedoch höchstens einige Tage halten, bevor sie zerstört wird.
Wenn Menschen in Entwicklungsländern aufgrund von Sperren, die erforderlich sind, um die Ausbreitung des Virus einzudämmen, arbeitslos bleiben, müssen sie weniger Geld ausgeben, was den Umsatz für Exporteure in Nordamerika, Europa und Ostasien verringert. Multinationale Unternehmen in fortgeschrittenen Ländern werden auch Schwierigkeiten haben, die erforderlichen Teile, Komponenten und Waren zu sichern.
Im Zentrum der Geschichte steht die Tatsache, dass der größte Teil des internationalen Handels keine fertigen Waren umfasst, sondern Teile, die von einem Land in ein anderes geliefert werden, um zu Produkten gefaltet zu werden. Von den Waren im Wert von 18 Billionen US-Dollar, die im vergangenen Jahr gehandelt wurden, machten sogenannte Zwischenprodukte nach Angaben der Organisation für wirtschaftliche Zusammenarbeit und Entwicklung 11 Billionen US-Dollar aus.
Die Studie stellt fest, dass die anhaltende Pandemie in armen Ländern wahrscheinlich am schlimmsten für Branchen ist, die besonders von Zulieferern auf der ganzen Welt abhängig sind, darunter Automobilindustrie, Textilindustrie, Bauwesen und Einzelhandel, in denen der Umsatz um mehr als 5 Prozent sinken könnte.
Die Ergebnisse ergänzen die Grundannahme, dass die Pandemie die Weltwirtschaft ungleicher als je zuvor machen wird, um eine komplizierende Schicht. Während dies wahr erscheint, könnte eine auffällige Form der Ungleichheit – der Zugang zu Impfstoffen – universelle Probleme aufwerfen.
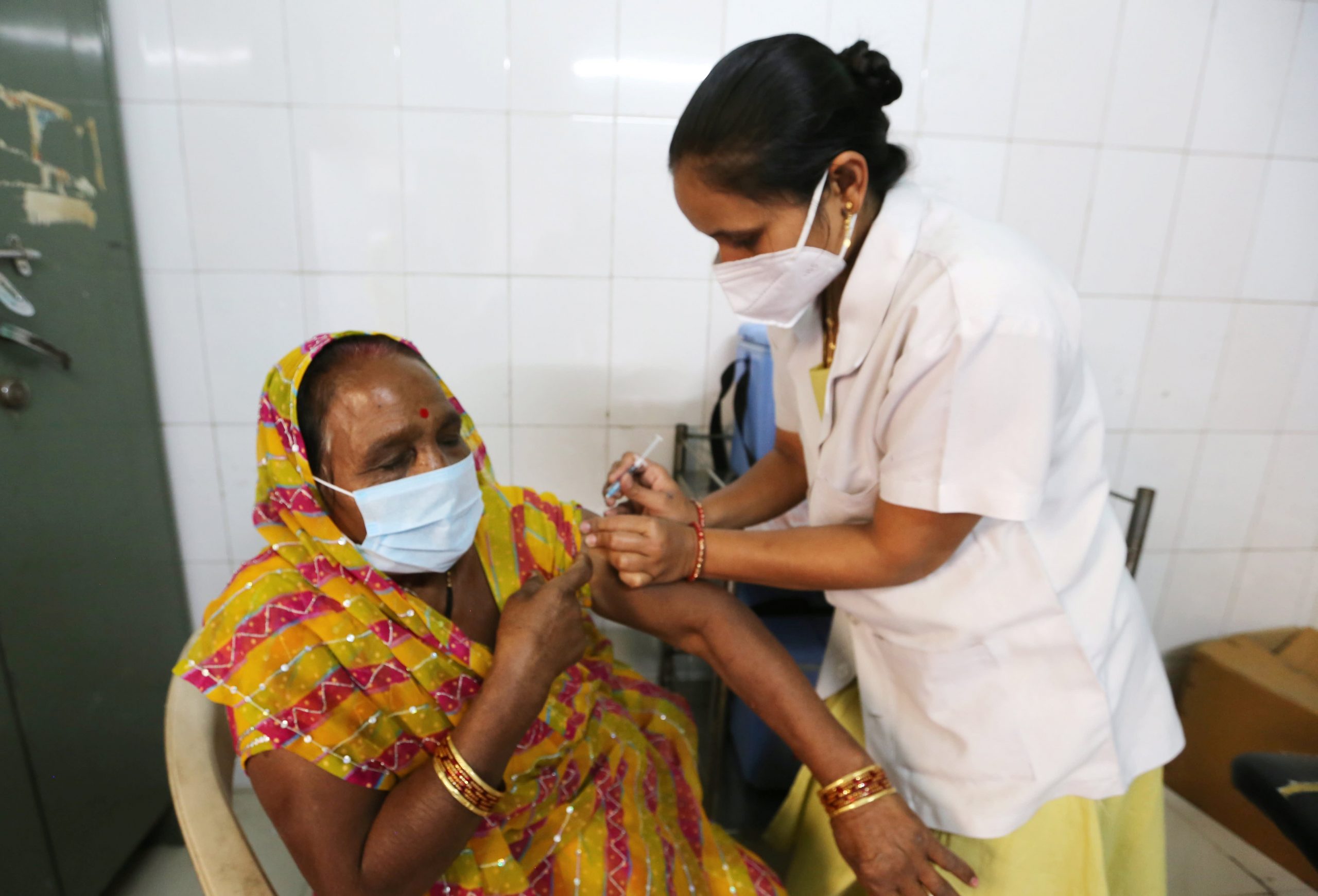
In einem außergewöhnlichen Beweis für die Innovationsfähigkeit der weltweit qualifiziertesten Wissenschaftler haben einige der führenden Pharmaunternehmen in einem kleinen Bruchteil der Zeit, die für möglich gehalten wurde, lebensrettende Impfstoffe hergestellt. Aber die reichsten Länder in Nordamerika und Europa haben Bestellungen für den größten Teil des Angebots abgeschlossen – genug, um das Zwei- und Dreifache ihrer Bevölkerung zu impfen – und die armen Länder haben sich bemüht, ihren Anteil zu sichern.
Viele Entwicklungsländer, von Bangladesch über Tansania bis Peru, werden wahrscheinlich bis 2024 warten müssen, bevor sie ihre Bevölkerung vollständig impfen können.
Die Initiative, arme Länder mit zusätzlichen Ressourcen zu versorgen, gewann mit dem Amtsantritt von Präsident Biden an Bedeutung. Die Trump-Administration hat nicht zur Sache beigetragen. Dr. Anthony S. Fauci, Chief Medical Officer von Herrn Biden für die Pandemie, kündigte umgehend an, dass die Vereinigten Staaten sich der Kampagne zum Austausch von Impfstoffen anschließen würden.
Im Gegensatz zu den Billionen Dollar, die Regierungen in reichen Ländern für die Rettung von Unternehmen und Arbeitnehmern ausgegeben haben, die durch den Gesundheitsnotstand und den wirtschaftlichen Abschwung geschädigt wurden, haben die Entwicklungsländer Schwierigkeiten, darauf zu reagieren.
Da Wanderarbeitnehmer aus armen Ländern während der Pandemie Arbeitsplätze verloren haben, konnten sie nicht so viel Geld nach Hause schicken, was Ländern, die sich auf diese sogenannten Überweisungen verlassen haben, wie den Philippinen, Pakistan und Bangladesch, einen schweren Schlag versetzt.
Die globale Rezession hat die Nachfrage nach Rohstoffen gedrosselt und Kupferproduzenten wie Sambia und die Demokratische Republik Kongo sowie von Öl abhängige Länder wie Angola und Nigeria dezimiert. Da die Fälle von Covid-19 stark angestiegen sind, hat dies den Tourismus gedrückt und Arbeitsplätze und Einnahmen in Thailand, Indonesien und Marokko gekostet.
Viele arme Länder sind mit Schuldenlasten in die Pandemie eingetreten, die einen Großteil ihrer Staatseinnahmen absorbierten und ihre Ausgaben für die Gesundheitsversorgung einschränkten. Private Gläubiger haben sich geweigert, an einem bescheidenen Programm zur Aussetzung der Schulden teilzunehmen, das von der Gruppe der 20 geschmiedet wurde. Die Weltbank und der Internationale Währungsfonds versprachen beide große Erleichterungen, brachten jedoch keine nennenswerten Dollars hervor.
Auch dies scheint sich zu ändern, da eine neue Führung Washington übernimmt. Die Trump-Regierung lehnte eine geplante Ausweitung der sogenannten Sonderziehungsrechte beim IWF um 500 Milliarden US-Dollar ab, einem Reservevermögen, das die Regierungen gegen harte Währungen eintauschen können. Der Aufstieg von Herrn Biden hat die Hoffnungen der Fondsmitglieder gestärkt, dass seine Verwaltung die Expansion unterstützen wird. Demokraten im Kongress – jetzt unter Kontrolle beider Kammern – haben Unterstützung für eine Maßnahme signalisiert, die das Finanzministerium zum Handeln zwingen würde.
In Hauptstädten wie Washington und Brüssel wurde die Diskussion über die Unterstützung der Entwicklungsländer jedoch moralisch gestaltet. Führer haben darüber diskutiert, wie viel sie übrig haben können, um den am wenigsten glücklichen Gemeinschaften des Planeten zu helfen, während sie sich hauptsächlich um ihre eigenen Leute kümmern.
Die Studie fordert diesen Rahmen heraus. Wenn es nicht darum geht, dass die Menschen in den Entwicklungsländern Zugang zu Impfstoffen erhalten, schaden die Staats- und Regierungschefs der reichsten Nationen ihrem eigenen Vermögen.
“Keine Wirtschaft, wie groß sie auch sein mag, wird gegen die Auswirkungen des Virus immun sein, bis die Pandemie überall ein Ende hat”, sagte John Denton, Generalsekretär der Internationalen Handelskammer. „Der Kauf von Impfstoffen für die Entwicklungsländer ist kein Akt der Großzügigkeit der reichsten Nationen der Welt. Es ist eine wesentliche Investition für die Regierungen, wenn sie ihre Binnenwirtschaft wiederbeleben wollen. “