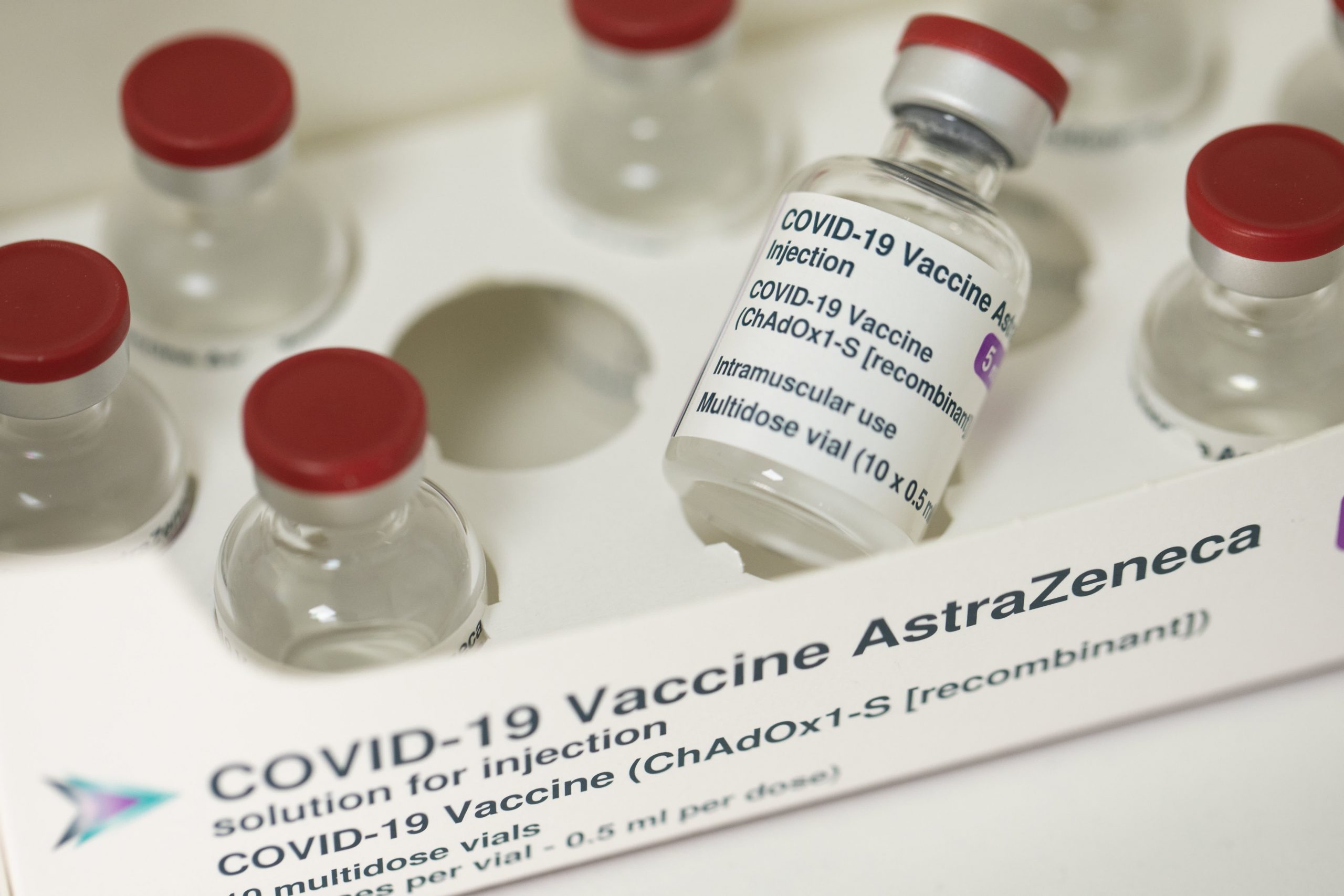
LONDON – This week’s announcement that the AstraZeneca shot, the workhorse of global vaccine adoption, was nearly 80 percent effective in a gold-standard American study was facilitated by the many countries that rely on it.
“If you get the call, get the bump,” urged UK Health Secretary Matt Hancock, part of a campaign by European lawmakers to calm people’s nerves with the shot after a recent security crisis.
But by Tuesday that campaign was off course, at least for the moment. For AstraZeneca, it appeared to be yet another episode of public relations whiplash, part of a string of recent mistakes and communications errors by the company that scientists said undermined efforts to sell people with one of the most effective and essential coronavirus vaccines.
In a highly unusual move, American health officials said Tuesday that the company’s report on the results of its US studies was not entirely accurate, suggesting that AstraZeneca used only the most favorable data to produce what appeared to be spectacular efficacy results.
These comments sparked new tensions between AstraZeneca and American officials, despite the company battling for coveted Food and Drug Administration approval. More urgently, however, she wrench the efforts of elected leaders around the world to restore confidence in a shot that, due to its low price and simple storage requirements, has rebuilt the backbone of many countries’ campaigns to end the pandemic have provided.
“It weakens confidence,” said Simon Clarke, associate professor of cell microbiology at the University of Reading. “If you pump things up and people don’t question it inappropriately, it undermines trust.”
Confidence in the vaccine had already fallen across Europe after it was recently reported that a very small number of recipients had developed unusual blood clots.
In France, Germany, Italy and Spain today, more people believe the vaccine is unsafe than it is safe. Polls have shown that this is a blow to a shot that remains the continent’s best hope of saving people’s lives amid a growing number of new infections. Millions of cans are used unused in refrigerators across the continent. Doctors report that some people stop injections because of concerns about side effects.
Despite the much worrying news about the vaccine, European and global regulators have found it safe and effective. In the UK alone, more than 11 million doses have been administered, almost all of which have no serious side effects. This led to hospital admissions and helped the country get out of a terrible wave of infections over the winter.
Even so, AstraZeneca’s US trial was eagerly awaited. It had been expected to be the largest of its kind for the shot and to give the cleanest, most complete picture of the vaccine’s effectiveness. American officials saw it as an irrefutable test of the vaccine’s performance.
And health officials around the world saw this as a crucial guide for their own rollouts: it would provide vital data on the elderly who weren’t as well represented in previous studies and a more accurate reading of the vaccine’s overall effectiveness. which from previous attempts had appeared lower than that of other leading shots.
As soon as AstraZeneca announced its results on Monday, stating that the vaccine had 79 percent effectiveness in preventing symptomatic Covid-19, lawmakers cited it as part of their fledgling efforts to build public confidence in the vaccine.
By Tuesday, scientists said, it appeared AstraZeneca had punched a hole in those efforts. Rather than clarifying questions about the shot, it had recalled communication issues that have haunted the company since last year, delaying the regulatory process in some regions, and causing hesitation among some recipients.
Updated
March 25, 2021, 8:30 a.m. ET
So far, according to the block, only 55 percent of the AstraZeneca doses shipped to the European Union have been placed in people’s arms, which is well below the rate of use for other vaccines. Around seven million cans are still in the fridge.
While some countries have given more than 70 percent of their doses, others are struggling to get them off the shelves: Germany and France have given about half of their AstraZeneca shipments, and Luxembourg has only given a third.
Scientists said such public dusting was extremely unusual between the American medical experts overseeing a study and the company sponsoring it.
“It’s usually done privately,” said Stephen Evans, professor of pharmacoepidemiology at the London School of Hygiene and Tropical Medicine, of any disagreement. “That is unprecedented in my opinion.”
In its first public comments, AstraZeneca said the results released on Monday mirrored US testing data through February 17. The preliminary assessment of more complete trial data found that “results are in line with interim analysis” said it would share more timely efficacy results within 48 hours.
Scientists said the problem might still turn out to be a technical matter that didn’t change their assessment of the vaccine. American officials did not suggest that security issues had been withheld, which was of great concern given concerns in Europe.
Even so, it quickly took the wind out of the sails of the European legislature’s public campaign to restore confidence in the shot developed with Oxford University. In the past few days, a number of political leaders, including British Prime Minister Boris Johnson and French Prime Minister Jean Castex, have received the vaccine itself to show people it is safe.
“I literally felt nothing,” Mr Johnson told reporters. “I don’t recommend it too much.”
Tuesday’s stumble was the latest in a series of mistakes that have created a troubled relationship between AstraZeneca and American and European regulators – and which scientists say has created unnecessary public confusion over a vaccine that appears to be highly effective.
In early September, the company silently halted its worldwide trials after a participant in the UK fell ill. But American regulators didn’t find out until the story became public. The company’s slowness in providing the FDA with evidence that its vaccine has not been linked to disease kept it on the ground for nearly seven weeks. AstraZeneca has announced that data will be exchanged in a timely manner.
By the end of November, the company was back at a high level: it published results of early clinical studies, including in the UK, that showed the vaccine was either 62 percent or 90 percent effective, depending on the type of dosage.
But even these results were quickly clouded by uncertainty. AstraZeneca later admitted that there was initially confusion about the dose of vaccine some study participants received, making the results difficult to interpret.
The UK, which has long advocated the home-grown vaccine, approved the shot in late December, citing earlier results from clinical trials. The European Union Medicines Agency did the same, but a month later.
EU officials said the delay was partly due to a back and forth between regulators and AstraZeneca over the quality of the data.
And even after the vaccine was approved, some European countries initially restricted it to younger people due to insufficient data on its effectiveness in the elderly. This problem was to be solved through the American process, in which the elderly were better represented.
Neither the European nor the UK regulators gave any indication on Tuesday that the problems with AstraZeneca’s American data would affect its launch there. These agencies relied on a separate dataset from non-American studies to approve the vaccine.
“We are in contact with the company regarding this additional information,” the European Medicines Agency said in a statement on Tuesday, “and the EMA will evaluate the relevant data as soon as the company provides it to us.”
Matina Stevis-Gridneff contributed to reporting from Brussels.