Florida Governor Ron DeSantis holds a press conference to announce the opening of a monoclonal antibody treatment center to help recover COVID-19 patients at Camping World Stadium in Orlando.
Paul Hennessy | LightRakete | Getty Images
Florida and Texas are opening free monoclonal antibody centers to treat a surge in Covid-19 patients in both states in the hopes that early intervention will help keep people out of hospitals and save more lives – even if they do The governors of both states are fighting local officials with mask and vaccination regulations.
Texas is building nine antibody infusion centers, Governor Greg Abbott announced on Friday, while Florida opened its fifth site on Wednesday. With the delta variant spike, coronavirus patients were occupied by more than 46% of Texas intensive care beds and more than half of Florida intensive care units as of Thursday, compared with 27% nationwide, according to the Department of Health and Social Affairs.
“What takes you to the hospital is the inflammation. People get inflammation in their lungs,” said Dr. Arturo Casadevall, Chair of Molecular Microbiology and Immunology at the Bloomberg School of Public Health at Johns Hopkins University, told CNBC in an interview. “So what these antibodies do is, if you give them to a patient early, they neutralize the virus.”
Abbott has firsthand experience of the treatment. His office announced Tuesday that he was receiving monoclonal antibody treatment from Regeneron after testing positive for Covid despite being fully vaccinated.
Although monoclonal antibodies like Regeneron and GlaxoSmithKline treatments are one of the few proven ways to fight the virus and reduce hospital stays, they were rarely used during the pandemic because they are awkward to administer. Monoclonal antibody treatments must be injected directly into the vein via an IV infusion, which requires time and dedicated medical staff, often using the same equipment reserved for chemotherapy patients.
The Food and Drug Administration issued emergency clearances to Regeneron’s treatment in November, saying it reduced hospital admissions for Covid “in patients at high risk for disease progression within 28 days of treatment.” GlaxoSmithKline just received emergency approval for its treatment with Vir Biotechnology in May and said it has reduced hospital stays and deaths in high-risk patients by about 85%.
The FDA approved both companies’ treatments for use in patients 12 years of age and older.
“Many patients who are examined by their doctors and referred for a monoclonal antibody infusion are less likely to be hospitalized,” said Teresa Farfan, spokeswoman for the Texas Division of Emergency Management, in an email to CNBC . “This will help ensure that resources are available in the hospitals to treat those with the most severe cases of the virus.”
Treatment centers couldn’t get there early enough as the Delta variant is driving cases to record highs in Florida. The state, which publishes its cases once a week on Fridays, last reported a record seven-day average of nearly 21,700 new infections, 12.6% more than a week ago, according to a CNBC analysis of data compiled by Hopkins.
Texas has been moving closer and closer to its record highs of more than 23,000 average cases per day in January in recent weeks, reporting a seven-day average of just over 15,400 new infections on Thursday, up from a seven-day average of around 3,000 a last month.
“Let me be very clear on this – both monoclonal and vaccines save lives,” said Christina Pushaw, spokeswoman for Florida Governor Ron DeSantis, in an email to CNBC. “They certainly aren’t mutually exclusive.”
More than 34% of the 50,706 registered inpatients in Florida have the coronavirus, as does over a quarter of the 51,337 registered inpatients in Texas, as measured Thursday. Abbott called 2,500 medical workers from across the country last week to help fight the virus and urged hospitals to build capacity by postponing election procedures.
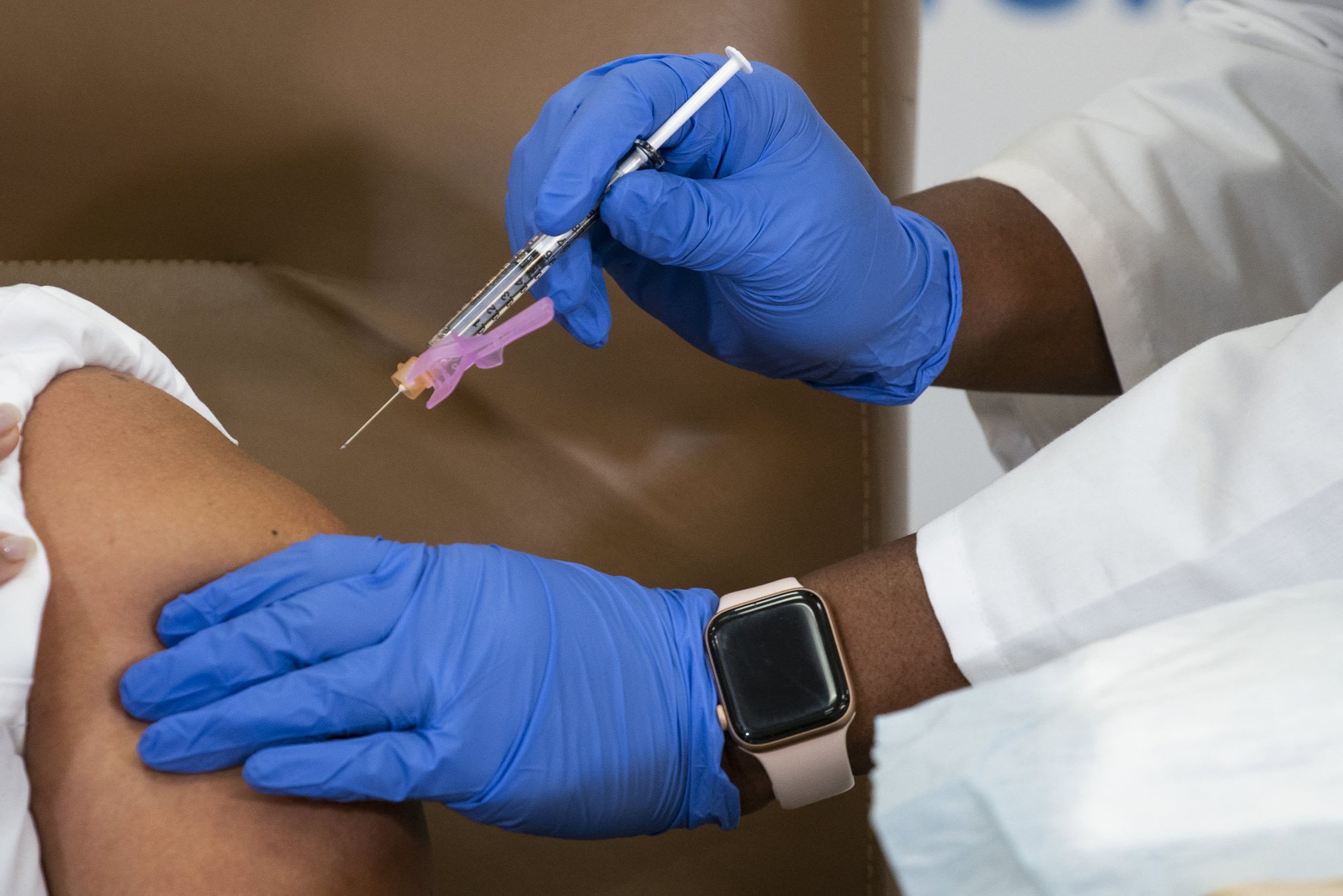
A box and vial of the Regeneron monoclonal antibody can be seen at a new COVID-19 treatment site opened by Florida Governor Ron DeSantis at Camping World Stadium in Orlando following a press conference.
Paul Hennessy | LightRakete | Getty Images
While both Abbott and DeSantis have urged residents to get vaccinated, they still strictly oppose mask or vaccination regulations, saying it violates personal freedoms. Republican governors have banned local governments and school districts from requiring face-covering. Abbott has threatened $ 1,000 fines for those who fail to comply, and DeSantis said it will withhold pay from educators who prescribe masks.
With many children returning to classrooms this fall, local officials are pushing back. Several school districts in both states have defied their governors’ orders and restored their mask mandates, with appeals courts in Dallas and San Antonio issuing injunctions last week to circumvent the ban.
The Texas Supreme Court on Sunday blocked the injunctions, sided with Abbott and prevented school districts from issuing their own guidelines. Local officials say they plan to continue fighting Abbott in court, and President Joe Biden on Wednesday directed the education secretary to intervene “to protect our children.”
“This includes using all of its regulators and, if necessary, taking legal action against governors who try to block and intimidate local school officials and educators,” said Biden.
Dr. Bruce Farber, chief of infectious diseases at Northwell Health in New York, said states that don’t allow schools to prescribe masks are at great risk this fall.
“These states are gambling as I see it,” he said in an interview. “By not allowing masking and preventing masking and leaving it to the parents, (they) are really playing with fire.”