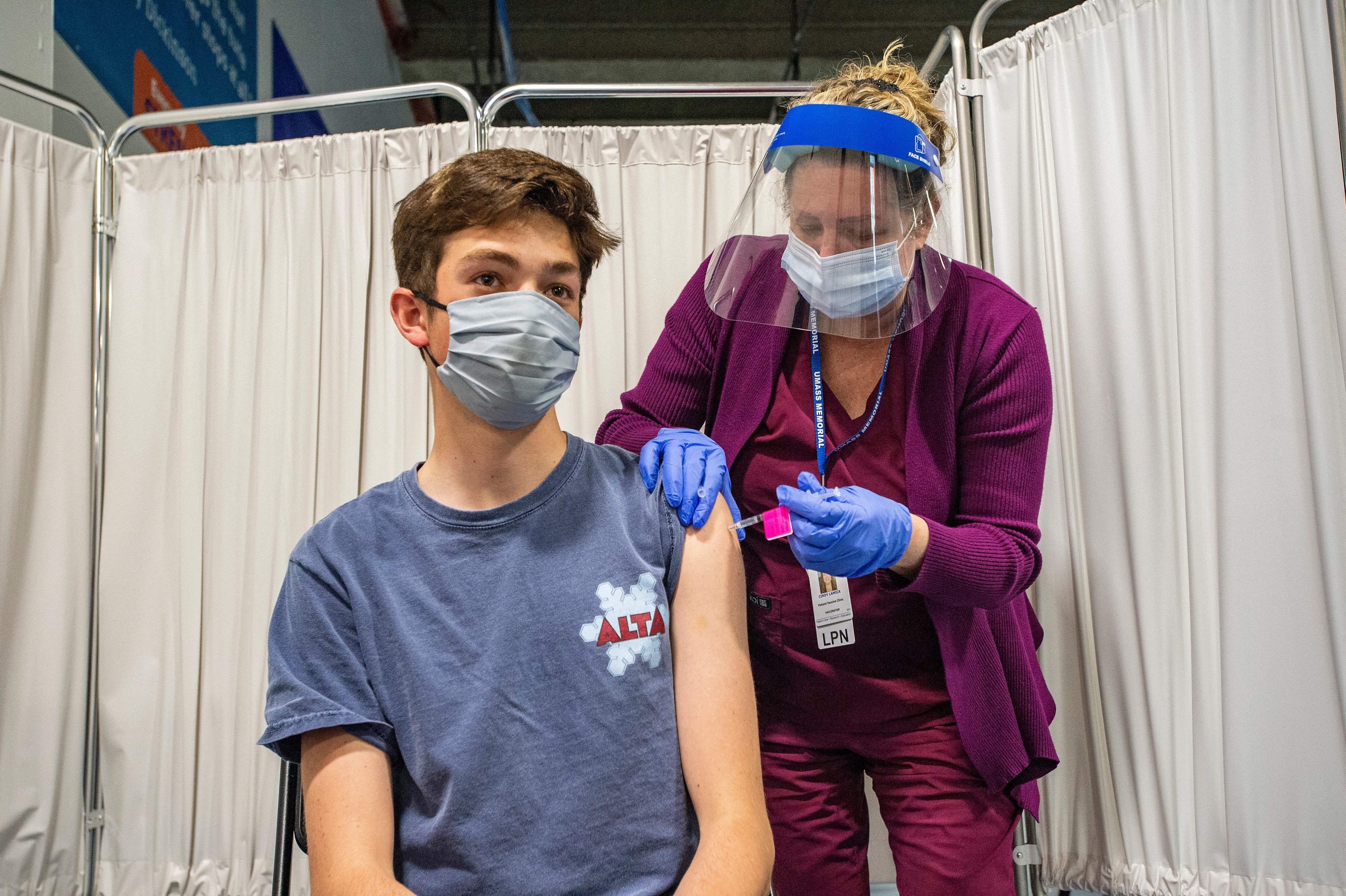
The federal government took one final step on Wednesday to bring Pfizer BioNTech coronavirus vaccine to teenagers in the United States, remove an obstacle to school reopening, and cheer millions of families tired of pandemic restrictions are.
An advisory committee of the Centers for Disease Control and Prevention voted The CDC director, Dr. Rochelle Walensky is expected to review the recommendations and approve them later on Wednesday.
“Getting Covid-19 vaccines approved for children ages 12-15 is an important step in removing barriers to vaccinating children of all ages,” said Dr. Yvonne Maldonado, who represents the American Academy of Pediatrics on the Federal Advisory Committee on Immunization Practices.
Many parents eagerly await the availability of vaccines for children, at least in part to expedite their return to school. About a third of eighth graders, usually 13 or 14 years old, still study completely remotely.
In some states, such as Maine, vaccination of teenagers has already started. Others plan to offer the vaccine as early as Thursday. There are nearly 17 million 12 to 15 year olds in the United States, which is 5.3 percent of the population.
Almost every state now has a flood of vaccine doses that could be quickly distributed to teenagers. The dose used to immunize adults is safe and effective for these adolescents too, as clinical studies have shown.
“Sometimes we lose the importance of children and adolescents in the midst of a pandemic – especially older adults are so much in focus,” said Dr. Grace Lee, Committee member and Professor of Pediatrics at Stanford University.
While the risk of serious illness in children is low compared to adults, the coronavirus has infected more than 1.5 million children and sent more than 13,000 to hospitals, according to the CDC, more than were hospitalized for flu in an average year
“It is currently one of the top ten causes of child death since the pandemic began,” noted Dr. Maldonado.
Young children are believed to be less likely to spread the virus than adults, but their ability to transmit it increases with age. Teenagers, especially in high school, can spread the virus just as easily as adults. Children aged 12-17 make up an increasing proportion of Covid cases in the country.
Vaccinating children should increase immunity levels in the US population and help reduce the number of cases.
“Any person with Covid-19 is giving the virus an opportunity to spread, further mutate and further expose our communities,” said Dr. Bill Gruber, Senior Vice President at Pfizer. “The decisions of the health authorities this week bring us one step closer to protecting young people and achieving herd protection.”
Pfizer announced in March that the vaccine appears to be at least as effective in 12-15 year olds as it is in older teenagers and adults. Apart from a slight increase in the frequency of fever, the shots also appeared to have comparable, mostly negligible side effects.
The company plans to monitor study participants for two years after the second dose to assess the long-term safety and effectiveness of the vaccine.
Updated
May 12, 2021, 4:58 p.m. ET
The Food and Drug Administration reviewed the clinical data and on Monday approved the Pfizer vaccine for use in these children. This allowed parents and children to wait weeks for a faster return to normal.
“While it is true that children are generally spared serious illnesses, the fact that they could not be vaccinated has created significant disruptions in their lives that have real developmental ramifications,” said Dr. Amesh Adalja, a senior scientist at the Johns Hopkins Center for Health Security. “By vaccinating this age cohort, these children can get back to their normal lives.”
In a speech at the White House on Wednesday, President Biden pointed out the benefits of the Covid vaccine for children 12 and older and said it was “safe, effective, easy, quick and free”.
“Starting tomorrow, more than 15,000 pharmacies will be ready to vaccinate this age group,” said Biden, adding that pharmacies would make it easier for teenagers to get the first shot in one location and a second shot in another location if needed.
Some experts have raised ethical concerns about vaccinating children who are at low risk for the virus, although healthcare workers and older adults remain at risk in many countries.
“If just thinking as a parent, if I had teenagers, I would probably love to vaccinate my children,” said Jennifer Nuzzo, an epidemiologist at the Johns Hopkins Center for Health Security.
Class disturbed
Updated May 5, 2021
The latest on how the pandemic is changing education.
But she added, “I am very concerned about a situation where the few countries in the world that had enough vaccines to protect their adults continue to hoard those vaccines for use in low-risk children.”
School reopenings have spiked across the country as parents, teachers, unions and school authorities worried about outbreaks. Research shows that children are largely spared serious illnesses and are not significant drivers of the spread of the coronavirus, as is the case with influenza.
“This misperception of risk will clearly divert vaccination priorities from the optimal strategic use of vaccines worldwide,” said Drs Adaliah.
The committee also recommended giving the Covid-19 vaccine along with other major vaccines that teenagers may have missed during the year. The agency had recommended waiting two weeks before and after immunization against Covid-19 before receiving other vaccines.
Parents’ reluctance can be the main hurdle. According to a recent survey by the Kaiser Family Foundation, more than 40 percent of parents of teenagers said that they would not vaccinate their children or would only vaccinate them if required by a school.
Some of these parents might change their minds as other children are safe to receive vaccines and resume personal schooling or return to team sports such as soccer and basketball that involve close contact, the researchers suggested.
Others can wait to meet school requirements. Public schools in all 50 states require certain vaccines, but officials may not be able to enforce compliance until the Pfizer BioNTech vaccine has received full FDA approval.
The vaccine now has an emergency approval. Pfizer has applied for full approval from the FDA, but this process is expected to take several months. Even after approval, students can opt out based on medical reasons or religious beliefs.
State and local leaders must make special efforts to reach children in low-income families or in color communities. Black and Hispanic adults have one of the lowest vaccination rates: by May 3, only 25 percent of blacks and 27 percent of Hispanics had been vaccinated, compared with 39 percent of whites.
In order to make the vaccine available to these communities, transport and storage of the cans must be facilitated. The Pfizer BioNTech vaccine can only be stored in standard refrigerators for five days. The companies plan to ship smaller packs for use in doctor’s offices and are developing a formulation that can be refrigerated for up to 10 weeks.
Pfizer and BioNTech plan to file applications for approval of the vaccine in children ages 2-11 in September.