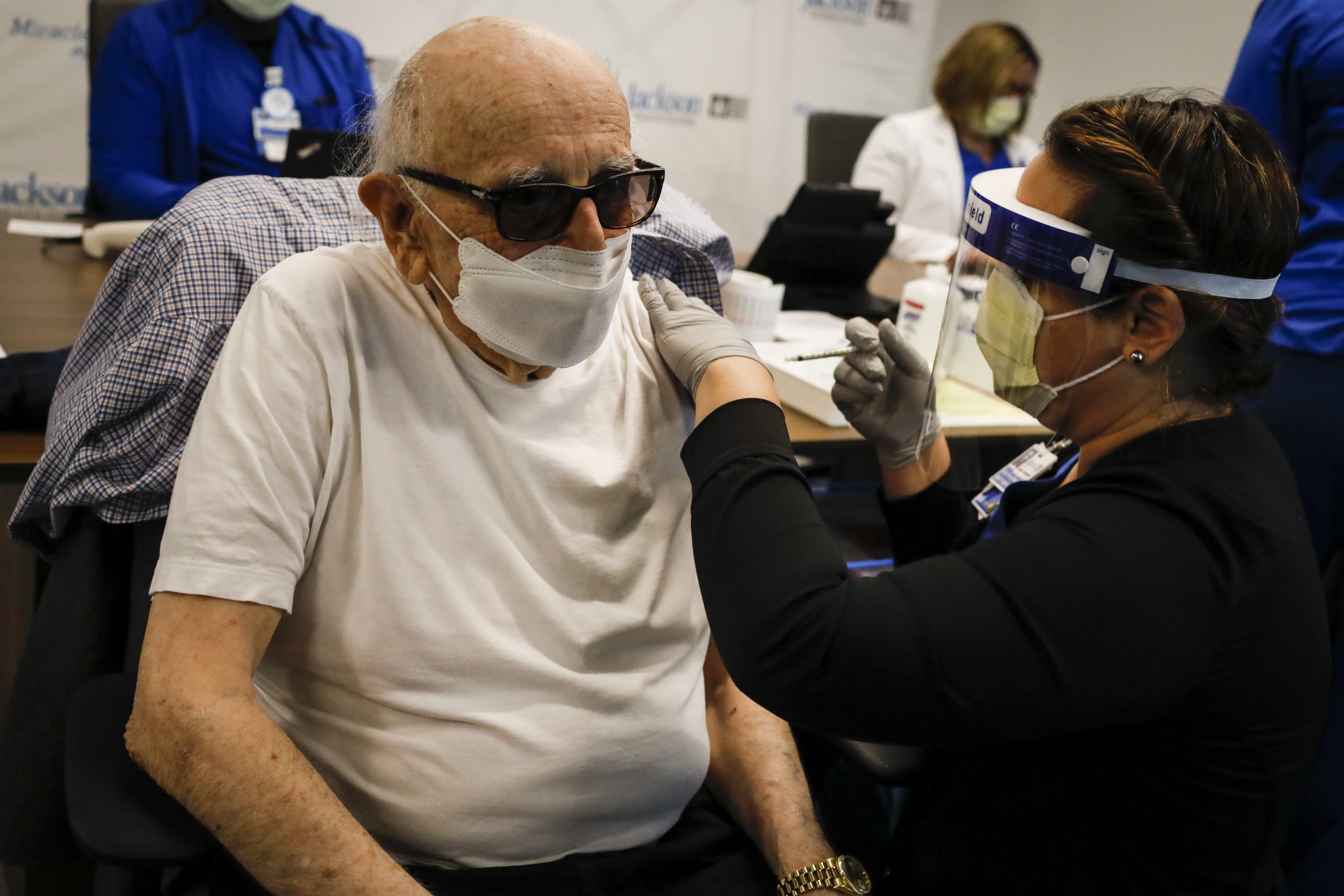
WASHINGTON – President Biden, faced with delayed vaccinations threatening his promise of near-normalcy through July 4th, revised its strategy to fight the pandemic on Tuesday, moving from mass vaccination sites to more local facilities to appeal to younger Americans and those who hesitate to get a shot.
In a speech at the White House, Mr Biden said he was launching a new phase in the fight against the coronavirus, with the aim of vaccinating at least 70 percent of adults at least partially by Independence Day, and with a personal appeal to all those who were not vaccinated: “That is Your decision. It’s life and death. “
After three months of tackling supply and distribution bottlenecks, the Biden government faces a problem the president deemed inevitable: many of those most likely to want to be vaccinated have already done so. Vaccination sites in stadiums that were once filled with truckloads of people looking for shots are closing, stating that once they ask for more vaccines, they won’t be able to use all of the doses the federal government wants to ship to them.
However, the government’s own health experts say an additional ten million Americans will need to be vaccinated before the infection rate is low enough to return to what many people consider normal life.
The administration now wants tens of thousands of pharmacies for people to take pictures. It has also ordered pop-up and mobile clinics, especially in rural areas, and plans to allocate tens of millions of dollars to outreach workers in the community to provide transportation and organize childcare for those in high-risk neighborhoods who are want to be vaccinated.
To build confidence in vaccines, federal officials plan to enlist the help of family doctors and other envoys who have trusted voices in their communities.
In a new effort to balance supply and demand, federal officials announced Tuesday that this vaccine would be considered part of a federal pool available to other states who so choose if they don’t get theirs in a given week would order full dose distribution to order more. So far, if states have not been able to order all of their allotted doses on a population basis, they could carry over that supply to the next week.
Mr Biden also announced a new federal website and phone number that will help people find the closest vaccination site. “We will make it easier than ever to get vaccinated,” he promised.
The government is hoping for a surge in vaccinations if the Food and Drug Administration approves the use of the Pfizer BioNTech vaccine for adolescents ages 12-15 as expected early next week. The president said adolescents are important in fighting the virus because while they are not as susceptible to serious illnesses, they can still get sick and infect others.
Experts say the United States may never achieve herd immunity. At this point the virus dies because there are no hosts to transmit it. And the president suggested the nation was still a long way from defeating the pandemic.
While the vast majority of seniors have been vaccinated, “we are still losing hundreds of Americans under 65 every week,” Biden said. “And many more get seriously ill at the same time from long distances.” He warned that the nation would vaccinate people in the fall.
Still, the president said that if 70 percent of the nation’s adults have had at least one vaccine by July 4th, “Americans will have taken a serious step toward a return to normal.”
To get there, the government needs to shift the focus from mass vaccination sites to doctor’s offices, pharmacies and other local facilities, and make a far more concerted effort to reach those who are reluctant to take pictures or just find out it’s too much trouble.
“We will move on,” said the president, optimistic that “most people will be convinced of the fact that their failure to receive the vaccine can lead to other people becoming sick and possibly dying.”
Updated
May 4, 2021, 3:12 p.m. ET
As of Tuesday, more than 106 million people in the United States were fully vaccinated and more than 56 percent of adults – or nearly 148 million people – had received at least one shot. This has contributed to sharp falls in infections, hospitalizations and deaths across all age groups, federal officials said.
Despite a flood of available doses, the rate of vaccination has dropped significantly in the past two and a half weeks. According to the Centers for Disease Control and Prevention, providers are currently delivering an average of about 2.19 million doses per day, down about 35 percent from the high of 3.38 million on April 13.
Mr Biden called for 160 million adults to be fully vaccinated by July 4 – an increase of 55 million people, or more than 50 percent. About 35 million more adults would have to get at least one shot to reach the president’s target of 70 percent of adults who are at least partially protected. While this next phase of the vaccination effort is “easier because I don’t have to put this massive logistical effort together,” said Mr Biden, “in the other sense it is more difficult, it is beyond my personal control.”
When asked if the United States would help other countries that are worse off, the president promised that by July 4th his administration will have “sent about 10 percent of what we have to other nations.” It wasn’t clear whether he was referring only to doses of AstraZeneca that are not approved for sale in the U.S. or to the country’s entire vaccine inventory. He also promised to act quickly “to get as many doses as possible from Moderna and Pfizer and export them around the world”.
So far, White House officials have stuck to formulas that assign vaccine doses to states by population and have been extremely reluctant to send doses of approved vaccines overseas. The government had been unwilling to move doses to states that could administer it faster, fearing that rural areas or underserved communities would lose to urban or richer areas where residents were more willing to get shots.
As the pace of vaccination slows down, officials have decided that the benefits of a loose system outweigh this risk.
States that want more than their allotment can ask for up to 50 percent more doses, officials said. States that do not claim all of their doses a week will not be penalized and will be able to claim their full allocations the next week, officials said.
The postponement makes little difference to some states that routinely obtained as many doses as the federal government was willing to ship. But it could help some states that can use more than the federal government has shipped.
White House press secretary Jen Psaki said Tuesday the move would give governors more flexibility. “Just a few weeks ago,” she said, “we were at a different stage in our vaccination efforts when supplies were more limited and states largely ordered at or near their full allotment.”
Virginia is a case in point. Last week, for the first time, the state didn’t order every dose it could have, said Dr. Danny Avula, the state vaccination coordinator.
Now he said, “If we can find ways to vaccinate a few people at a time, supply will exceed demand across the state, and work will be much slower and more difficult.” Dr. Avula said the change will “be very helpful to the few states that still have localized areas of high demand.”
Low demand states like Arkansas may find their allotted doses shipped to an alternate location. Arkansas has so far only used 69 percent of the doses it has been given, data shows. Last week, a health ministry spokeswoman said the state had not ordered cans from the federal government. Just over a third of Arkansas adults have received at least one dose, one of the lowest in the country.
Ms. Psaki said the government is working with states to find out which settings make the most sense at this point in the vaccination campaign.
“We’re constantly evaluating the best delivery mechanisms,” she said, “and if something isn’t the most effective, we will make changes.”
Mr Biden suggested that general practitioners and pediatricians play a key role in promoting the vaccination program, as do other community figures. If the Pfizer vaccine is approved for teenagers, the administration plans to make it immediately available to them in about 20,000 pharmacies that participate in the federal vaccination program.
However, some cans are being shipped direct to pediatricians so “parents and their children can talk to their GP and get the shot from a provider they trust most,” the president said. Dr. Vivek Murthy, the surgeon general, said last week that “80 percent of people who try to decide on a vaccine say they want to speak to their doctor about that decision – and we heard that loud and clear. ”