Norwegian Cruise Line threatens to keep its ships out of Florida ports after the state enacted laws prohibiting companies from requesting proof of Covid-19 vaccination in exchange for services.
The company, which plans to launch its first cruises to the Caribbean and Europe in the summer and fall, offers limited capacity trips and requires all guests and crew to be vaccinated for bookings by at least the end of October.
During a quarterly earnings call Thursday, Frank Del Rio, managing director of Norwegian Cruise Line, said the issue had been discussed with Florida Governor Ron DeSantis, a Republican. Mr Del Rio said if the cruise line had to skip the ports of Florida, it could operate from other states or the Caribbean.
“We definitely hope it doesn’t come to that,” said Mr Del Rio. “Everyone wants to operate from Florida. It’s a very lucrative market. “
The conflict between Norwegian Cruise Line and Florida is one of the many that is likely to surface when it comes to how states and companies go about whether or not proof of vaccination is required. While some states are not yet taking a position on companies that require vaccines, others are already using such protocols.
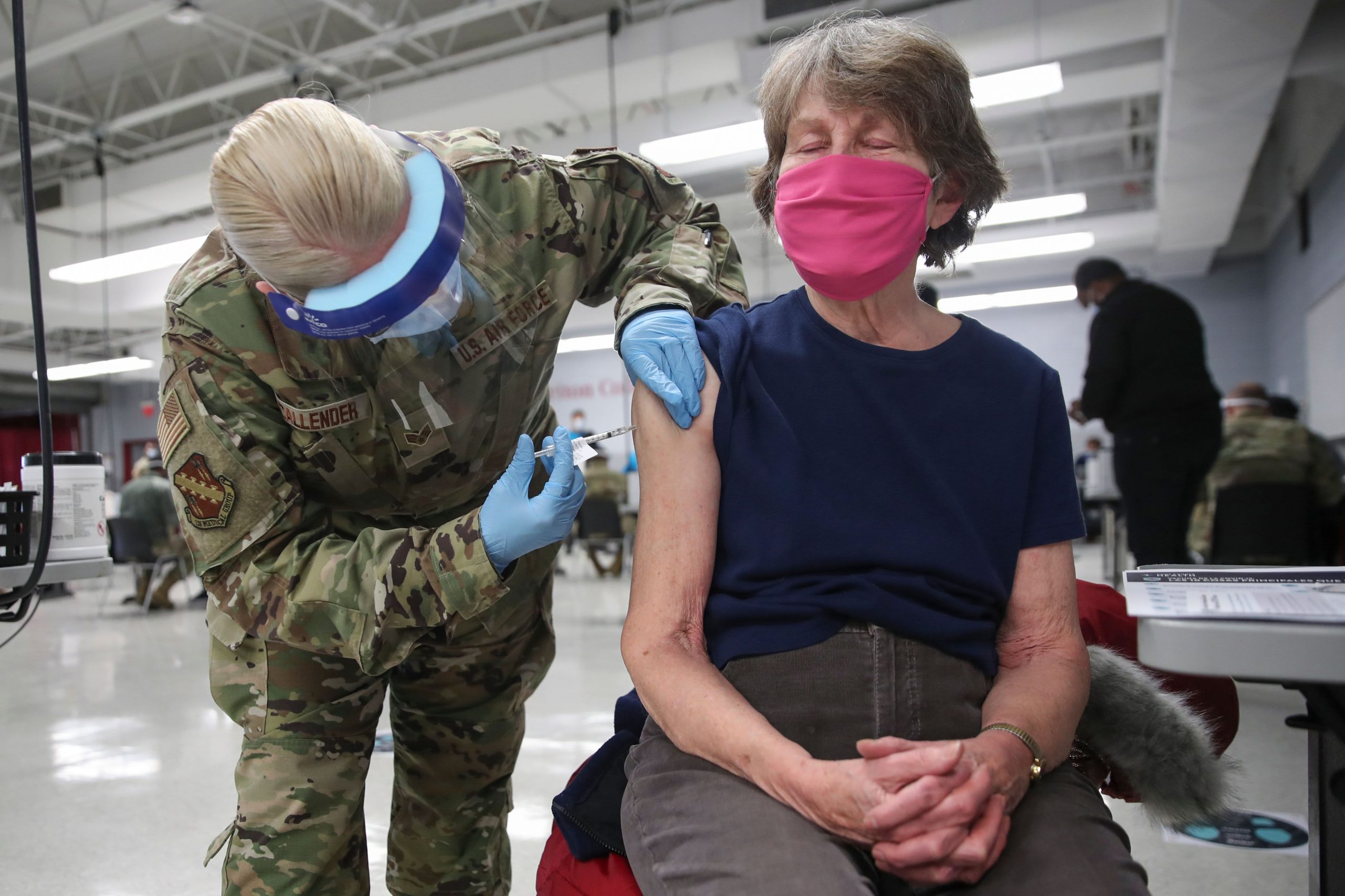
At many New York events, such as Major League Baseball and National Basketball Association games, state health and safety guidelines require fans to provide a vaccination certificate or negative coronavirus test within 72 hours of attending.
“We hope this has not become legal or political football,” said Del Rio on the conference call.
Norwegian Cruise Line is headquartered in Florida, along with Royal Caribbean Cruises and Carnival Corporation. According to an economic analysis prepared for the Cruise Lines International Association last year, around 60 percent of all US cruise ships in 2019 came from ports in Florida.
In a business update on Thursday, Norwegian Cruise Line announced that bookings for the first half of 2022 were seeing “robust future demand” that was “well ahead” of 2019 bookings. By the end of the first quarter of 2021, the company announced a pre-sale of $ 1.3 billion in tickets.
Florida law not only prohibits companies from providing evidence of vaccination, but also prevents state and local authorities from closing personal learning companies or schools unless there is a hurricane emergency.
Updated
May 8, 2021, 2:21 p.m. ET
“I have refused to take the same approach as other lockdown governors,” DeSantis said in a statement on Monday when he signed the bill. “Florida protects your personal choice about vaccinations and no company or government agency can deny you services based on your choice.”
His office did not immediately respond to a request for comment on Saturday and Norwegian Cruise Line could not be reached for comment.
“We hope everyone is pushing in the same direction, which means we want to safely resume the cruise, especially at the beginning,” said Del Rio on the call for winners. “In six months or a year, things could be different.”
The latest guidelines from the Centers for Disease Control and Prevention allow cruise lines to conduct “simulated voyages” with volunteer passengers to see how cruise lines can safely resume operations with measures such as testing and potential quarantines.
The CDC requires cruise ships to complete test runs before they can be cleared for sailing with passengers this summer.
“It is not possible for cruises to be an activity without risk for the spread of Covid-19,” the CDC said this week. “While cruises always pose some risk to the transmission of Covid-19, CDC is committed to ensuring that cruise ship passenger operations are carried out in a way that protects crew, passengers and port personnel.”
The latest guidelines recommend but do not require that cruise ship travelers and crew members receive a vaccine when it is available to them.
Speaking at this week’s call for a prize, Mr. Del Rio said Norwegian Cruise Line had submitted a proposal to the CDC requiring vaccine detection from all crew members and passengers.
It’s unclear how much business Norwegian Cruise Line could lose by avoiding Florida ports. Of the dozen of ports listed on its website, Norwegian Cruise Line has Florida ports in Tampa, Miami, and Key West.
Mr Del Rio said “pent-up demand” helped fill bookings quickly.
“I believe it’s the # 1 destination for Americans in the Caribbean,” said Del Rio. “Who knows? This ship could prove so profitable there that it will never return to US waters.”