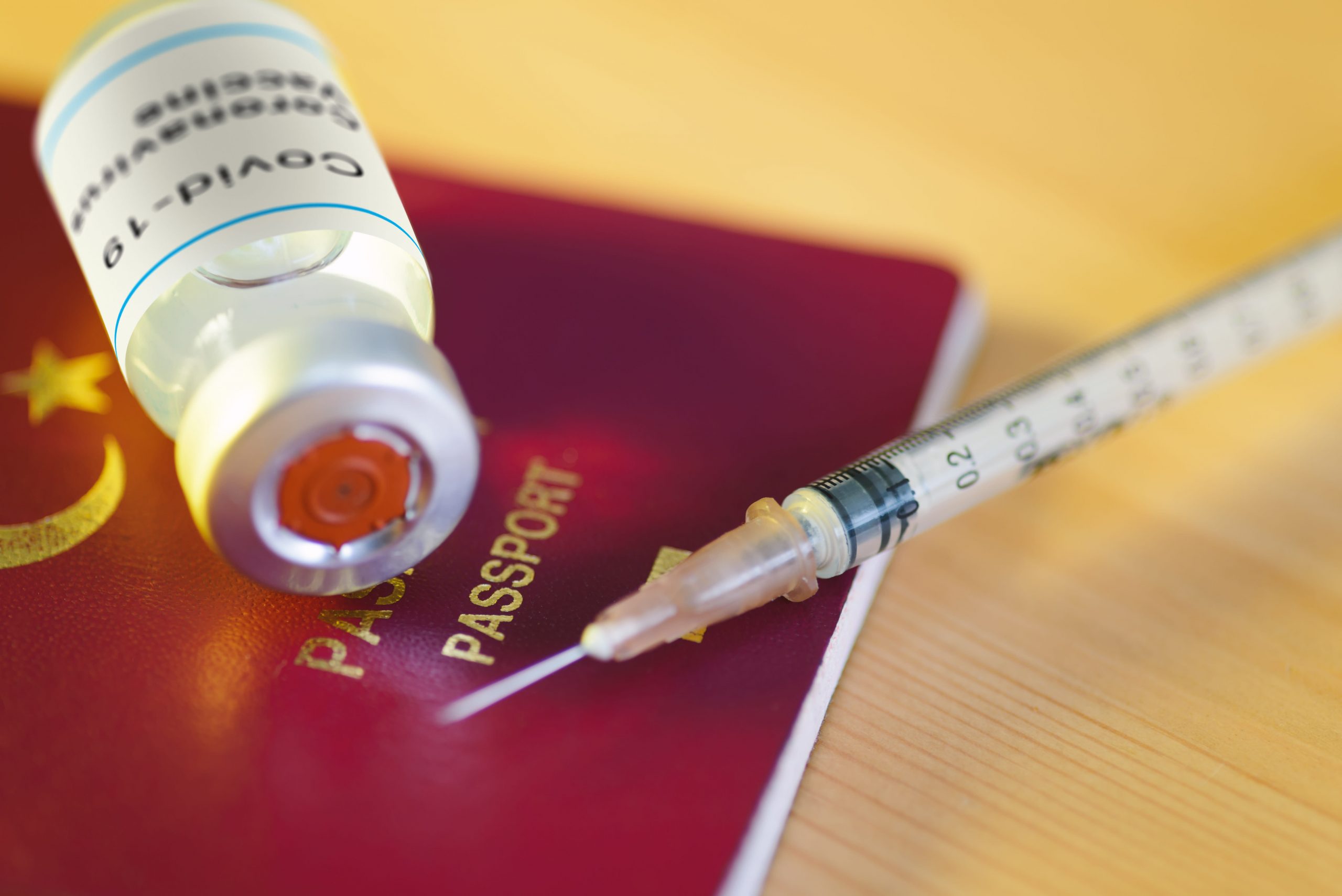
As the adoption of vaccinations gains momentum around the world, attention is now turning to vaccines of a different kind: vaccination records.
Last week, the International Air Transport Association announced the launch of its new digital passport as a “way forward” for the resumption of quarantine-free international travel.
The app, which is being tested by 30 airlines, enables governments and airlines to collect, access and share encrypted information related to the passengers’ Covid-19 test and vaccination status before departure.
The International Chamber of Commerce and the World Economic Forum have developed similar apps – ICC AOKpass and CommonPass – that travelers can use to electronically document their medical status. Countries like Denmark and Sweden are introducing their own health passports, and even tech giants want to join.
What are digital health passes and will they allow a return to heaven this year?
What is a vaccination certificate?
A vaccination card, also known as a digital health card, is digital documentation that a person has been vaccinated against a virus, in this case Covid.
Stored on a phone or digital wallet, the data is usually presented as a QR code and can also indicate whether a person has tested negative for a virus.
Digital health passports are tested to validate people’s Covid-19 test and vaccination status.
Maskot | Getty Images
Such documentation is not unprecedented. For decades, people traveling to certain countries have had to show physical “yellow cards” as proof of vaccination against diseases such as cholera, yellow fever and rubella.
However, this is the first time the industry has advocated an electronic alternative to improve auditability and bypass some of the delays caused by paper peers.
“Imagine the scene when 180,000 people present a piece of paper that needs review and validation,” said Mike Tansey, general manager at Accenture, referring to the number of daily passengers at Changi Airport in Singapore prior to Covid.
Do we need digital health passes to travel?
Tansey, who leads Accenture’s APAC Travel and Hospitality division, has worked with several major airlines on their digital health passport strategies, including three in the US and several in the Asia-Pacific region.
He told CNBC’s Global Traveler that these plans have “accelerated” since the vaccine was launched, and the need for such IDs is clear to him.
The obvious answer is yes we do.
Mike Tansey
General Manager, Travel and Hospitality, Accenture
“The obvious answer is’ yes,” Tansey said when asked if we would need digital health cards to resume the trip.
He called debates a “red herring”.
“Governments may not say you have to have one, but the effects of not will be so ridiculous that travel isn’t worth it,” he said, referring to extensive testing and “draconian” quarantines.
What are the security concerns?
Tansey is not alone. Other experts agree that digital health passes are the fastest, most effective way to resume international travel.
Jase Ramsey, professor of management at Florida Gulf Coast University’s Lutgert College of Business, agreed that the likelihood of adoption was “very high”. However, he noted that concerns about security and personal information could make consumers less willing to use digital health passports than their physical alternatives.
“As with any app that stores health records, there are privacy and fraud concerns,” said Ramsey.
Vaccination records electronically store medical information that is displayed as a QR code.
da-kuk | E + | Getty Images
Accredify is a Singapore-based document accreditation firm whose technology is used as part of the Singapore government mandated Covid-19 pre-travel health screening. It is claimed that the appeal of digital accreditation systems – like its own, which is based on the blockchain – is that they are tamper-proof and therefore cannot be forged.
“Medical documents that are privately and securely stored in the app are only accessible to users, so they can decide who and when to share their medical records with,” a spokesman said via email.
Traveler resistance can be overrated. A recent study by travel news website The Vacationer found that 73.6% of Americans surveyed would use a Covid health pass or app so airlines and border agencies can check their vaccination status and test results.
What are the challenges for health passports?
The success of digital health passports depends on the effectiveness of vaccines. Little is known about whether vaccines prevent the spread of Covid, although research is currently being carried out.
The World Health Organization has urged caution with health cards and urged the authorities and tour operators not to introduce proof of vaccination as a condition for international travel.
The effectiveness of vaccines in preventing transmission is not yet clear and global vaccine supplies are limited.
speaker
World health organization
“This is because the effectiveness of vaccines in preventing transmission is not yet clear and global vaccine supplies are limited,” said a WHO spokesman.
Coordinating the various existing and pending vaccination records in the market and ensuring that users’ certifications are linked to verified and approved medical facilities will prove to be a major challenge.
“For vaccine passports to be an internationally practical tool, there must be a standardized platform that crosses all borders – like the current passport system,” said Dr. Harry Severance, Assistant Professor at Duke University School of Medicine.
WHO works with agencies such as the International Air Transport Association and the International Civil Aviation Organization to develop standards for digital vaccination cards. It added that its position on health passports “will evolve as the evidence for existing and new Covid-19 vaccines is updated”.
What about the social impact?
Add to this, of course, the social, legal and political implications of a system based on unequal global access to vaccines and technology.
According to the WHO, around 3.6 billion people worldwide cannot access the Internet and more than 1.1 billion cannot officially prove their identity. For many, paper IDs remain essential.
Access to vaccinations is still far from fair around the world
Luis Alvarez | DigitalVision | Getty Images
“People from different countries, regions or communities may not have access to vaccines or Covid-19 tests,” said Dr. Sharona Hoffman, a bioethics professor at Case Western Reserve University’s School of Medicine, noting that low-income countries may not get vaccinations until 2023 or beyond. “Policies that prevent them from traveling or using other services could be discriminatory and exacerbate socio-economic disparities.”
Such systems could also set a precedent for other groups that are also eager to reopen, such as B. Restaurants and Event Venues. In fact, Israel has already created a “green passport” to give vaccinated citizens access to public venues.
This week, some US states decided to lift mask mandates, which could exacerbate this problem.
“As one community moves in this direction, many, many more will follow. As decisions like this are made across the country, you may find that ‘carding’ vaccines becomes the standard,” Severance said.
What could this mean for the future of travel?
Ultimately, resumption of international travel will depend as much on countries’ willingness to reopen as it does on existing travel verification technology.
In the Asia-Pacific region, where borders remain largely closed to tourists, governments may lean towards bilateral agreements or “travel bubbles” with select neighbors before opening further, Accenture’s Tansey said.
An internationally recognized system of health passports … will potentially enable us to survive an impending pandemic.
Harry Severance
Duke University School of Medicine
“The The reality … is that we are six months away from meaningful air travel, “he said.” Agreements are only made with one or two locations at a time. “
However, with much of the technology in place and society moving towards an increasingly digitized future, today’s developments in digital health passports could better prepare the travel industry – and society – for potential turmoil.
“As we evolve into an internationally recognized system of monitoring health passports (or) etc, it will be a facet of a downstream preparedness system that may allow us to survive an impending pandemic that may have worse dynamics than Covid- 19. ” “said Severance.