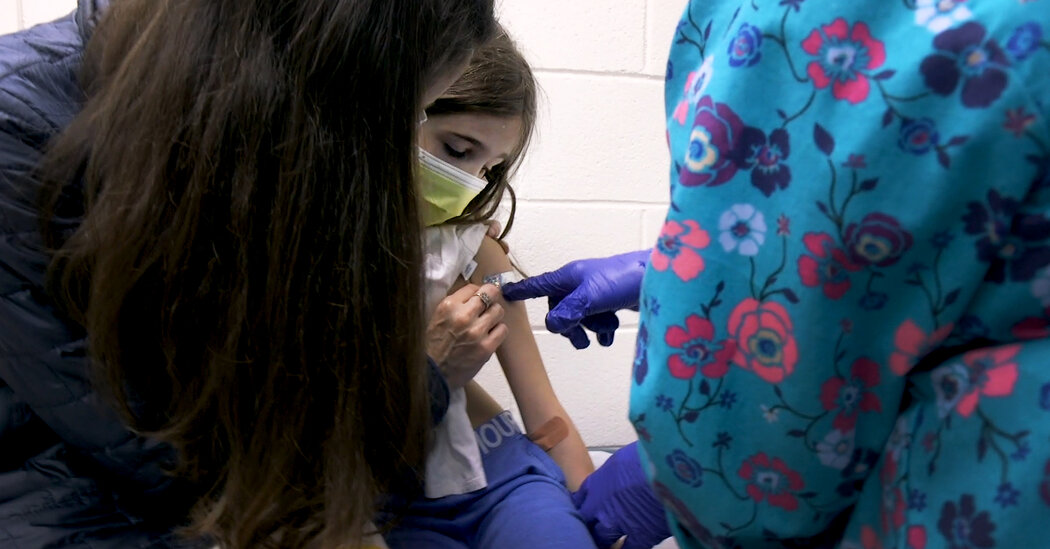
Pfizer has started testing its Covid-19 vaccine in children under the age of 12. This is an important step in reducing the pandemic. The first participants in the study, a pair of 9-year-old twin girls, were vaccinated on Wednesday at Duke University in North Carolina.
Results of the study are expected in the second half of the year, and the company hopes to vaccinate younger children early next year, said Sharon Castillo, a spokeswoman for the drug company.
Moderna is also starting testing its vaccine in children aged six months to 12 years. Both companies have tested their vaccines in children 12 years and older and expect these results in the next few weeks.
AstraZeneca began testing its vaccine in children six months and older last month. Johnson & Johnson has announced plans to extend the vaccine trials to young children after assessing performance in older children.
Immunizing children will help schools reopen and end the pandemic, said Dr. Emily Erbelding, an infectious disease doctor at the National Institutes of Health who oversees the testing of Covid-19 vaccines in specific populations.
An estimated 80 percent of the population may need to be vaccinated for the United States to achieve herd immunity, the threshold above which the coronavirus can no longer infect people. Some adults may refuse to be vaccinated, while others may not produce a robust immune response.
Children under the age of 18 make up about 23 percent of the US population. Even if the vast majority of adults choose vaccines, “herd immunity may be difficult to achieve without vaccinating children,” said Dr. Erbelding.
Pfizer originally announced that it would wait for data from older children before starting trials of its vaccine in children under the age of 12. “We were encouraged, however, by the data from the group of 12-15 year olds,” said Ms. Castillo, who did not elaborate on results so far.
Scientists will test three doses of the Pfizer vaccine – 10, 20, and 30 micrograms – in 144 children. Each dose is assessed first in children aged 5 to 11 years, then in children aged 2 to 4 years, and finally in the youngest group aged six months to 2 years.
After determining the most effective dose, the company will test the vaccine on 4,500 children. Approximately two-thirds of the participants are randomly selected to receive two doses 21 days apart. The remainder received two placebo injections of saline solution. The researchers will study the children’s immune response in blood drawn seven days after the second dose.
Updated
March 25, 2021, 2:39 p.m. ET
“It sounds like a good plan, and it’s exciting to see another Covid-19 vaccine drive studies in children,” said Dr. Kristin Oliver, pediatrician and vaccine expert at Mount Sinai Hospital in New York.
Dr. Oliver said that about half of the parents she sees in the office eagerly await vaccines and even volunteer their children for clinical trials, while the rest are skeptical because comparatively few children get seriously ill with coronavirus infection .
Both parent groups will benefit from knowing exactly how safe and effective the vaccines are in children, she said.
Children make up 13 percent of all reported cases in the United States. More than 3.3 million children tested positive for the virus, at least 13,000 were hospitalized and at least 260 died, noted Dr. Yvonne Maldonado, who represents the American Academy of Pediatrics on the Federal Advisory Board on Immunization Practices.
The figures do not fully capture the damage to the health of children. “We don’t know how a Covid infection will affect the long term,” said Dr. Maldonado.
Other vaccines have helped fight many terrible teething problems that can cause long-term complications. She added, “For some of us who have seen this, we don’t want to go back to that time.”
Children are often more responsive to vaccines than adults, and infants and young children in particular can have a high fever. All side effects are likely to appear soon after the shot, within the first week, and certainly within the first few weeks, experts have said.
Some vaccines are only tested on animals before being studied in children and must be carefully monitored for side effects.
“But that’s a little different because we’ve already had tens of millions of people with these vaccines,” said Dr. Maldonado. “So there is more confidence to give this vaccine to children.”
Some experts suggested that the Food and Drug Administration may need up to six months of safety data from studies in children before the Covid-19 vaccines are approved. However, a spokeswoman said the agency did not expect safety data to support approval of the vaccines for six months.
The Pfizer BioNTech vaccine is approved for children ages 16-18, and approval for that age group was based on just two months of safety data, she said.
Parents will want to know how the companies and the FDA plan to monitor and disclose the side effects of the vaccines and how long they will pursue study participants after the vaccines are approved, said Dr. Oliver.
“I think everyone has learned that,” she said. “The more transparent you can be, the better.”